Molecular iodine--an expedient reagent for oxidative aromatization reactions of alpha,beta-unsaturated cyclic compounds
- PMID: 20032894
- PMCID: PMC6254800
- DOI: 10.3390/molecules14125308
Molecular iodine--an expedient reagent for oxidative aromatization reactions of alpha,beta-unsaturated cyclic compounds
Abstract
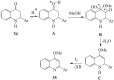
Prompted by the scant attention paid by published literature reviews to the applications of molecular iodine in oxidative aromatization reactions, we decided to review methods developed to-date involving iodine as an oxidant to promote aromatization of alpha,beta-unsaturated cyclic compounds.
Figures
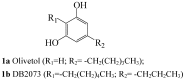
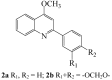
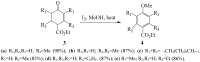


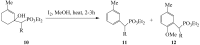


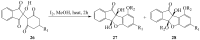

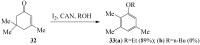
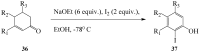

Similar articles
-
Reagent-controlled oxidative aromatization in iodocyclization: switchable access to dihydropyrazoles and pyrazoles.Org Lett. 2010 Aug 6;12(15):3506-9. doi: 10.1021/ol101365x. Org Lett. 2010. PMID: 20614873
-
[Novel iodocyclization method based on the controlling of oxidative aromatization].Yakugaku Zasshi. 2011;131(9):1323-7. doi: 10.1248/yakushi.131.1323. Yakugaku Zasshi. 2011. PMID: 21881306 Review. Japanese.
-
Iodocyclization of hydroxylamine derivatives based on the control of oxidative aromatization leading to 2,5-dihydroisoxazoles and isoxazoles.J Org Chem. 2011 May 6;76(9):3438-49. doi: 10.1021/jo200407b. Epub 2011 Apr 7. J Org Chem. 2011. PMID: 21449558
-
Synthesis of 2-azaindolizines by using an iodine-mediated oxidative desulfurization promoted cyclization of N-2-pyridylmethyl thioamides and an investigation of their photophysical properties.Org Lett. 2006 Nov 23;8(24):5621-4. doi: 10.1021/ol0623623. Org Lett. 2006. PMID: 17107087
-
Iodine-catalyzed oxidative coupling reactions utilizing C - H and X - H as nucleophiles.Chem Asian J. 2015 Apr;10(4):806-23. doi: 10.1002/asia.201403248. Epub 2015 Feb 12. Chem Asian J. 2015. PMID: 25683644 Review.
Cited by
-
The two-step mechanochemical synthesis of porphyrins.Faraday Discuss. 2014;170:59-69. doi: 10.1039/c3fd00140g. Faraday Discuss. 2014. PMID: 25406483 Free PMC article.
-
Ethyl 3-(2,4-dioxocyclohexyl)propanoate as a novel precursor for N-substituted 4,4a,5,6-tetrahydroquinoline-2,7(1H,3H)-diones and their corresponding 3,4-dihydro-7-hydroxyquinolin-2(1H)-ones and 7-hydroxyquinolin-2(1H)-ones synthesis.Mol Divers. 2016 Feb;20(1):29-40. doi: 10.1007/s11030-015-9643-x. Epub 2015 Nov 4. Mol Divers. 2016. PMID: 26537729
References
-
- Horning E.C., Horning M.C. Aromatization Studies. IV. Palladium dehydrogenation of arylcyclohexenones to phenols. J. Am. Chem. Soc. 1947;69:1359–1361. doi: 10.1021/ja01198a035. - DOI
-
- Horning E.C., Horning M.C., Walker G.N. Aromatization Studies. VIII. Catalytic dehydrogenation of di- and trialkylcyclohexenones to phenols. J. Am. Chem. Soc. 1949;71:169–171. doi: 10.1021/ja01169a044. - DOI
-
- Bondon D., Pietrasanta Y., Pucci B. New methods of aromatization of cyclohexenones contained in polycyclic systems. Tetrahedron Lett. 1977:821–824. doi: 10.1016/S0040-4039(01)92765-0. - DOI
-
- Yamaguchi S., Yamamoto K., Ueda T., Morikawa T., Kawase Y. A new preparative method of 4-hydroxybenzofuran-2-carboxylic acid derivatives. Bull. Chem. Soc. Jpn. 1989;62:4066–4068. doi: 10.1246/bcsj.62.4066. - DOI
-
- Grieco P.A., Marinovic N. Rhodium-catalyzed migration of double bonds: preparation of substituted phenols and anilines. Tetrahedron Lett. 1978;29:2545–2548. doi: 10.1016/S0040-4039(01)94822-1. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

