Regulatory function of cytomegalovirus-specific CD4+CD27-CD28- T cells
- PMID: 20034645
- PMCID: PMC2823847
- DOI: 10.1016/j.virol.2009.11.038
Regulatory function of cytomegalovirus-specific CD4+CD27-CD28- T cells
Abstract
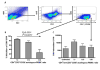
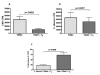
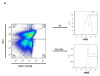
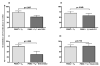
CMV infection is characterized by high of frequencies of CD27-CD28- T cells. Here we demonstrate that CMV-specific CD4+CD27-CD28- cells are regulatory T cells (TR). CD4+CD27-CD28- cells sorted from CMV-stimulated PBMC of CMV-seropositive donors inhibited de novo CMV-specific proliferation of autologous PBMC in a dose-dependent fashion. Compared with the entire CMV-stimulated CD4+ T-cell population, higher proportions of CD4+CD27-CD28- TR expressed FoxP3, TGFbeta, granzyme B, perforin, GITR and PD-1, lower proportions expressed CD127 and PD1-L and similar proportions expressed CD25, CTLA4, Fas-L and GITR-L. CMV-CD4+CD27-CD28- TR expanded in response to IL-2, but not to CMV antigenic restimulation. The anti-proliferative effect of CMV-CD4+CD27-CD28- TR significantly decreased after granzyme B or TGFbeta inhibition. The CMV-CD4+CD27-CD28- TR of HIV-infected and uninfected donors had similar phenotypes and anti-proliferative potency, but HIV-infected individuals had higher proportions of CMV-CD4+CD27-CD28- TR. The CMV-CD4+CD27-CD28- TR may contribute to the downregulation of CMV-specific and nonspecific immune responses of CMV-infected individuals.
Copyright 2009 Elsevier Inc. All rights reserved.
Figures
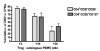




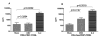
Similar articles
-
Cytomegalovirus infection in HIV-infected and uninfected individuals is characterized by circulating regulatory T cells of unconstrained antigenic specificity.PLoS One. 2017 Jul 6;12(7):e0180691. doi: 10.1371/journal.pone.0180691. eCollection 2017. PLoS One. 2017. PMID: 28683106 Free PMC article.
-
Regulatory T cells generated during cytomegalovirus in vitro stimulation of mononuclear cells from HIV-infected individuals on HAART correlate with decreased lymphocyte proliferation.Virology. 2006 Sep 1;352(2):408-17. doi: 10.1016/j.virol.2006.04.035. Epub 2006 Jun 19. Virology. 2006. PMID: 16782163
-
PD-1 analysis on CD28(-) CD27(-) CD4 T cells allows stimulation-independent assessment of CMV viremic episodes in transplant recipients.Am J Transplant. 2013 Dec;13(12):3132-41. doi: 10.1111/ajt.12480. Epub 2013 Oct 22. Am J Transplant. 2013. PMID: 24148296
-
CMV-specific T-cells and CD27-CD28-CD4+ T-cells for assignment of cytomegalovirus (CMV) status in adults awaiting organ transplant.J Clin Virol. 2019 Jun;115:37-42. doi: 10.1016/j.jcv.2019.03.014. Epub 2019 Mar 25. J Clin Virol. 2019. PMID: 30959325
-
Effect of Exercise on Acute Senescent Lymphocyte Counts: A Systematic Review and Meta-Analysis.Gerontology. 2022;68(9):961-975. doi: 10.1159/000520528. Epub 2022 Jan 14. Gerontology. 2022. PMID: 35034018
Cited by
-
Regulatory T cells and the risk of CMV end-organ disease in patients with AIDS.J Acquir Immune Defic Syndr. 2014 May 1;66(1):25-32. doi: 10.1097/QAI.0000000000000095. J Acquir Immune Defic Syndr. 2014. PMID: 24378728 Free PMC article.
-
Understanding the mechanism of action of cytomegalovirus-induced regulatory T cells.Virology. 2020 Aug;547:1-6. doi: 10.1016/j.virol.2020.05.001. Epub 2020 May 10. Virology. 2020. PMID: 32442104 Free PMC article.
-
IL-10-Secreting CD8+ T Cells Specific for Human Cytomegalovirus (HCMV): Generation, Maintenance and Phenotype.Pathogens. 2022 Dec 13;11(12):1530. doi: 10.3390/pathogens11121530. Pathogens. 2022. PMID: 36558866 Free PMC article.
-
B and T Cell Phenotypic Profiles of African HIV-Infected and HIV-Exposed Uninfected Infants: Associations with Antibody Responses to the Pentavalent Rotavirus Vaccine.Front Immunol. 2018 Jan 19;8:2002. doi: 10.3389/fimmu.2017.02002. eCollection 2017. Front Immunol. 2018. PMID: 29403482 Free PMC article.
-
Generation, maintenance and tissue distribution of T cell responses to human cytomegalovirus in lytic and latent infection.Med Microbiol Immunol. 2019 Aug;208(3-4):375-389. doi: 10.1007/s00430-019-00598-6. Epub 2019 Mar 20. Med Microbiol Immunol. 2019. PMID: 30895366 Free PMC article. Review.
References
-
- Adeegbe D, Bayer AL, Levy RB, Malek TR. Cutting edge: allogeneic CD4+CD25+Foxp3+ T regulatory cells suppress autoimmunity while establishing transplantation tolerance. J Immunol. 2006;176 (12):7149–53. - PubMed
-
- Almanzar G, Schwaiger S, Jenewein B, Keller M, Herndler-Brandstetter D, Wurzner R, Schonitzer D, Grubeck-Loebenstein B. Long-term cytomegalovirus infection leads to significant changes in the composition of the CD8+ T-cell repertoire, which may be the basis for an imbalance in the cytokine production profile in elderly persons. J Virol. 2005;79 (6):3675–83. - PMC - PubMed
-
- Bekker V, Bronke C, Scherpbier HJ, Weel JF, Jurriaans S, Wertheim-van Dillen PM, van Leth F, Lange JM, Tesselaar K, van Baarle D, Kuijpers TW. Cytomegalovirus rather than HIV triggers the outgrowth of effector CD8+CD45RA+CD27− T cells in HIV-1-infected children. Aids. 2005;19 (10):1025–34. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

