Phosphorylation of the F(1)F(o) ATP synthase beta subunit: functional and structural consequences assessed in a model system
- PMID: 20035080
- PMCID: PMC2835499
- DOI: 10.1161/CIRCRESAHA.109.214155
Phosphorylation of the F(1)F(o) ATP synthase beta subunit: functional and structural consequences assessed in a model system
Abstract
Rationale: We previously discovered several phosphorylations to the beta subunit of the mitochondrial F(1)F(o) ATP synthase complex in isolated rabbit myocytes on adenosine treatment, an agent that induces cardioprotection. The role of these phosphorylations is unknown.
Objective: The present study focuses on the functional consequences of phosphorylation of the ATP synthase complex beta subunit by generating nonphosphorylatable and phosphomimetic analogs in a model system, Saccharomyces cerevisiae.
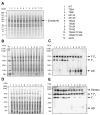
Methods and results: The 4 amino acid residues with homology in yeast (T58, S213, T262, and T318) were studied with respect to growth, complex and supercomplex formation, and enzymatic activity (ATPase rate). The most striking mutant was the T262 site, for which the phosphomimetic (T262E) abolished activity, whereas the nonphosphorylatable strain (T262A) had an ATPase rate equivalent to wild type. Although T262E, like all of the beta subunit mutants, was able to form the intact complex (F(1)F(o)), this strain lacked a free F(1) component found in wild-type and had a corresponding increase of lower-molecular-weight forms of the protein, indicating an assembly/stability defect. In addition, the ATPase activity was reduced but not abolished with the phosphomimetic mutation at T58, a site that altered the formation/maintenance of dimers of the F(1)F(o) ATP synthase complex.
Conclusions: Taken together, these data show that pseudophosphorylation of specific amino acid residues can have separate and distinctive effects on the F(1)F(o) ATP synthase complex, suggesting the possibility that several of the phosphorylations observed in the rabbit heart can have structural and functional consequences to the F(1)F(o) ATP synthase complex.
Figures


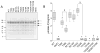


Similar articles
-
Subunit f of the yeast mitochondrial ATP synthase: topological and functional studies.J Bioenerg Biomembr. 1999 Apr;31(2):85-94. doi: 10.1023/a:1005407525915. J Bioenerg Biomembr. 1999. PMID: 10449235
-
Manipulations in the peripheral stalk of the Saccharomyces cerevisiae F1F0-ATP synthase.J Biol Chem. 2011 Mar 25;286(12):10155-62. doi: 10.1074/jbc.M110.213447. Epub 2011 Jan 21. J Biol Chem. 2011. PMID: 21257750 Free PMC article.
-
Consequences of the pathogenic T9176C mutation of human mitochondrial DNA on yeast mitochondrial ATP synthase.Biochim Biophys Acta. 2010 Jun-Jul;1797(6-7):1105-12. doi: 10.1016/j.bbabio.2009.12.022. Epub 2010 Jan 4. Biochim Biophys Acta. 2010. PMID: 20056103 Free PMC article.
-
Assembly of F0 in Saccharomyces cerevisiae.Biochim Biophys Acta. 2009 Jan;1793(1):108-16. doi: 10.1016/j.bbamcr.2008.07.001. Epub 2008 Jul 11. Biochim Biophys Acta. 2009. PMID: 18672007 Free PMC article. Review.
-
Rotational catalysis in proton pumping ATPases: from E. coli F-ATPase to mammalian V-ATPase.Biochim Biophys Acta. 2012 Oct;1817(10):1711-21. doi: 10.1016/j.bbabio.2012.03.015. Epub 2012 Mar 20. Biochim Biophys Acta. 2012. PMID: 22459334 Review.
Cited by
-
Dephosphorylation of cardiac proteins in vitro - a matter of phosphatase specificity.Proteomics. 2012 Apr;12(7):973-8. doi: 10.1002/pmic.201100116. Proteomics. 2012. PMID: 22522803 Free PMC article.
-
Chronic Activation of AMPK Induces Mitochondrial Biogenesis through Differential Phosphorylation and Abundance of Mitochondrial Proteins in Dictyostelium discoideum.Int J Mol Sci. 2021 Oct 28;22(21):11675. doi: 10.3390/ijms222111675. Int J Mol Sci. 2021. PMID: 34769115 Free PMC article.
-
Targeting regulation of ATP synthase 5 alpha/beta dimerization alleviates senescence.Aging (Albany NY). 2022 Jan 30;14(2):678-707. doi: 10.18632/aging.203858. Epub 2022 Jan 30. Aging (Albany NY). 2022. PMID: 35093936 Free PMC article.
-
Probing the Energetic Metabolism of Resting Cysts under Different Conditions from Molecular and Physiological Perspectives in the Harmful Algal Blooms-Forming Dinoflagellate Scrippsiella trochoidea.Int J Mol Sci. 2021 Jul 7;22(14):7325. doi: 10.3390/ijms22147325. Int J Mol Sci. 2021. PMID: 34298944 Free PMC article.
-
Hippocampal versus cortical deletion of cholinergic receptor muscarinic 1 in mice differentially affects post-translational modifications and supramolecular assembly of respiratory chain-associated proteins, mitochondrial ultrastructure, and respiration: implications in Alzheimer's disease.Front Cell Dev Biol. 2023 May 24;11:1179252. doi: 10.3389/fcell.2023.1179252. eCollection 2023. Front Cell Dev Biol. 2023. PMID: 37293125 Free PMC article.
References
-
- Liem DA, Honda HM, Zhang J, Woo D, Ping P. Past and present course of cardioprotection against ischemia-reperfusion injury. J Appl Physiol. 2007;103:2129–36. - PubMed
-
- Murphy E, Steenbergen C. Preconditioning: the mitochondrial connection. Annu Rev Physiol. 2007;69:51–67. - PubMed
-
- Arrell DK, Elliott ST, Kane LA, Guo Y, Ko YH, Pedersen PL, Robinson J, Murata M, Murphy AM, Marban E, Van Eyk JE. Proteomic analysis of pharmacological preconditioning: novel protein targets converge to mitochondrial metabolism pathways. Circ Res. 2006;99:706–14. - PubMed
-
- Ala-Rami A, Ylitalo KV, Hassinen IE. Ischaemic preconditioning and a mitochondrial KATP channel opener both produce cardioprotection accompanied by F1F0-ATPase inhibition in early ischaemia. Basic Res Cardiol. 2003;98:250–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

