Signal strength and signal duration define two distinct aspects of JNK-regulated axon stability
- PMID: 20035736
- PMCID: PMC2845820
- DOI: 10.1016/j.ydbio.2009.12.016
Signal strength and signal duration define two distinct aspects of JNK-regulated axon stability
Abstract
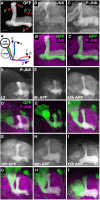
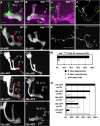
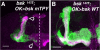
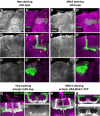
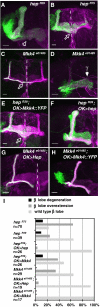
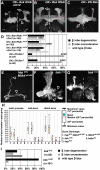
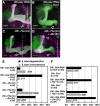
Signaling proteins often control multiple aspects of cell morphogenesis. Yet the mechanisms that govern their pleiotropic behavior are often unclear. Here we show activity levels and timing mechanisms determine distinct aspects of Jun N-terminal kinase (JNK) pathway dependent axonal morphogenesis in Drosophila mushroom body (MB) neurons. In the complete absence of Drosophila JNK (Basket), MB axons fail to stabilize, leading to their subsequent degeneration. However, with a partial loss of Basket (Bsk), or of one of the upstream JNK kinases, Hemipterous or Mkk4, these axons overextend. This suggests that Bsk activity prevents axons from destabilizing, resulting in degeneration and overextension beyond their terminal targets. These distinct phenotypes require different threshold activities involving the convergent action of two distinct JNK kinases. We show that sustained Bsk signals are essential throughout development and act additively but are dispensable at adulthood. We also suggest that graded Bsk inputs are translated into AP-1 transcriptional outputs consisting of Fos and Jun proteins.
Copyright 2009 Elsevier Inc. All rights reserved.
Figures

Similar articles
-
DISCO interacting protein 2 determines direction of axon projection under the regulation of c-Jun N-terminal kinase in the Drosophila mushroom body.Biochem Biophys Res Commun. 2017 May 20;487(1):116-121. doi: 10.1016/j.bbrc.2017.04.028. Epub 2017 Apr 8. Biochem Biophys Res Commun. 2017. PMID: 28396149
-
Degeneration of Injured Axons and Dendrites Requires Restraint of a Protective JNK Signaling Pathway by the Transmembrane Protein Raw.J Neurosci. 2019 Oct 23;39(43):8457-8470. doi: 10.1523/JNEUROSCI.0016-19.2019. Epub 2019 Sep 6. J Neurosci. 2019. PMID: 31492772 Free PMC article.
-
The Ste20 kinase misshapen regulates both photoreceptor axon targeting and dorsal closure, acting downstream of distinct signals.Mol Cell Biol. 2000 Jul;20(13):4736-44. doi: 10.1128/MCB.20.13.4736-4744.2000. Mol Cell Biol. 2000. PMID: 10848599 Free PMC article.
-
Jun N-terminal kinase signaling makes a face.Fly (Austin). 2016 Oct;10(4):195-203. doi: 10.1080/19336934.2016.1207012. Epub 2016 Jul 6. Fly (Austin). 2016. PMID: 27384866 Free PMC article. Review.
-
Molecular signaling how do axons die?Adv Genet. 2011;73:185-217. doi: 10.1016/B978-0-12-380860-8.00005-7. Adv Genet. 2011. PMID: 21310297 Review.
Cited by
-
The making of the Drosophila mushroom body.Front Physiol. 2023 Jan 13;14:1091248. doi: 10.3389/fphys.2023.1091248. eCollection 2023. Front Physiol. 2023. PMID: 36711013 Free PMC article. Review.
-
Axon Termination, Pruning, and Synaptogenesis in the Giant Fiber System of Drosophila melanogaster Is Promoted by Highwire.Genetics. 2017 Mar;205(3):1229-1245. doi: 10.1534/genetics.116.197343. Epub 2017 Jan 18. Genetics. 2017. PMID: 28100586 Free PMC article.
-
The equilibrium between antagonistic signaling pathways determines the number of synapses in Drosophila.PLoS One. 2017 Sep 11;12(9):e0184238. doi: 10.1371/journal.pone.0184238. eCollection 2017. PLoS One. 2017. PMID: 28892511 Free PMC article.
-
Novel function of N-acetyltransferase for microtubule stability and JNK signaling in Drosophila organ development.Proc Natl Acad Sci U S A. 2021 Jan 26;118(4):e2010140118. doi: 10.1073/pnas.2010140118. Proc Natl Acad Sci U S A. 2021. PMID: 33479178 Free PMC article.
-
Pathway crosstalk enables cells to interpret TGF-β duration.NPJ Syst Biol Appl. 2018 May 28;4:18. doi: 10.1038/s41540-018-0060-5. eCollection 2018. NPJ Syst Biol Appl. 2018. PMID: 29872541 Free PMC article.
References
-
- Agnes F., Suzanne M., Noselli S. The Drosophila JNK pathway controls the morphogenesis of imaginal discs during metamorphosis. Development. 1999;126:5453–5462. - PubMed
-
- Bettencourt-Dias M., Giet R., Sinka R., Mazumdar A., Lock W.G., Balloux F., Zafiropoulos P.J., Yamaguchi S., Winter S., Carthew R.W., Cooper M., Jones D., Frenz L., Glover D.M. Genome-wide survey of protein kinases required for cell cycle progression. Nature. 2004;432:980–987. - PubMed
-
- Bjorkblom B., Ostman N., Hongisto V., Komarovski V., Filen J.J., Nyman T.A., Kallunki T., Courtney M.J., Coffey E.T. Constitutively active cytoplasmic c-Jun N-terminal kinase 1 is a dominant regulator of dendritic architecture: role of microtubule-associated protein 2 as an effector. J. Neurosci. 2005;25:6350–6361. - PMC - PubMed
-
- Bjorklund M., Taipale M., Varjosalo M., Saharinen J., Lahdenpera J., Taipale J. Identification of pathways regulating cell size and cell-cycle progression by RNAi. Nature. 2006;439:1009–1013. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

