Voltage-dependent anion channel 1-based peptides interact with Bcl-2 to prevent antiapoptotic activity
- PMID: 20037155
- PMCID: PMC2825399
- DOI: 10.1074/jbc.M109.082990
Voltage-dependent anion channel 1-based peptides interact with Bcl-2 to prevent antiapoptotic activity
Abstract
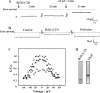
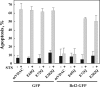
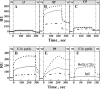
The antiapoptotic proteins of the Bcl-2 family are expressed at high levels in many types of cancer. However, the mechanism by which Bcl-2 family proteins regulate apoptosis is not fully understood. Here, we demonstrate the interaction of Bcl-2 with the outer mitochondrial membrane protein, voltage-dependent anion channel 1 (VDAC1). A direct interaction of Bcl-2 with bilayer-reconstituted purified VDAC was demonstrated, with Bcl-2 decreasing channel conductance. Expression of Bcl-2-GFP prevented apoptosis in cells expressing native but not certain VDAC1 mutants. VDAC1 sequences and amino acid residues important for interaction with Bcl-2 were defined through site-directed mutagenesis. Synthetic peptides corresponding to the VDAC1 N-terminal region and selected sequences bound specifically, in a concentration- and time-dependent manner, to immobilized Bcl-2, as revealed by the real-time surface plasmon resonance. Moreover, expression of the VDAC1-based peptides in cells over-expressing Bcl-2 prevented Bcl-2-mediated protection against staurosporine-induced apoptotic cell death. Similarly, a cell-permeable VDAC1-based synthetic peptide was also found to prevent Bcl-2-GFP-mediated protection against apoptosis. These results point to Bcl-2 as promoting tumor cell survival through binding to VDAC1, thereby inhibiting cytochrome c release and apoptotic cell death. Moreover, these findings suggest that interfering with the binding of Bcl-2 to mitochondria by VDAC1-based peptides may serve to potentiate the efficacy of conventional chemotherapeutic agents.
Figures

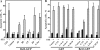

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

