The descending corticocollicular pathway mediates learning-induced auditory plasticity
- PMID: 20037578
- PMCID: PMC3634157
- DOI: 10.1038/nn.2466
The descending corticocollicular pathway mediates learning-induced auditory plasticity
Abstract
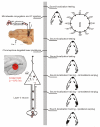
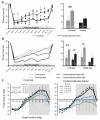
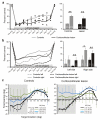
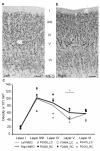
Descending projections from sensory areas of the cerebral cortex are among the largest pathways in the brain, suggesting that they are important for subcortical processing. Although corticofugal inputs have been shown to modulate neuronal responses in the thalamus and midbrain, the behavioral importance of these changes remains unknown. In the auditory system, one of the major descending pathways is from cortical layer V pyramidal cells to the inferior colliculus in the midbrain. We examined the role of these neurons in experience-dependent recalibration of sound localization in adult ferrets by selectively killing the neurons using chromophore-targeted laser photolysis. When provided with appropriate training, animals normally relearn to localize sound accurately after altering the spatial cues available by reversibly occluding one ear. However, this ability was lost after eliminating corticocollicular neurons, whereas normal sound-localization accuracy was unaffected. The integrity of this descending pathway is therefore critical for learning-induced localization plasticity.
Figures
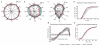

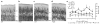

References
-
- Dahmen JC, King AJ. Learning to hear: plasticity of auditory cortical processing. Curr. Opin. Neurobiol. 2007;17:456–464. - PubMed
-
- Edeline JM, Weinberger NM. Thalamic short-term plasticity in the auditory system: associative returning of receptive fields in the ventral medial geniculate body. Behav. Neurosci. 1991;105:618–639. - PubMed
-
- Suga N, Ma X. Multiparametric corticofugal modulation and plasticity in the auditory system. Nat. Rev. Neurosci. 2003;4:783–794. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

