Toxicity of xanthene food dyes by inhibition of human drug-metabolizing enzymes in a noncompetitive manner
- PMID: 20041016
- PMCID: PMC2778353
- DOI: 10.1155/2009/953952
Toxicity of xanthene food dyes by inhibition of human drug-metabolizing enzymes in a noncompetitive manner
Abstract
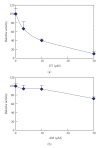
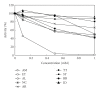
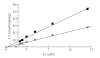
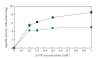
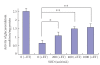
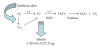
The synthetic food dyes studied were rose bengal (RB), phroxine (PL), amaranth, erythrosine B (ET), allura red, new coccine, acid red (AR), tartrazine, sunset yellow FCF, brilliant blue FCF, and indigo carmine. First, data confirmed that these dyes were not substrates for CYP2A6, UGT1A6, and UGT2B7. ET inhibited UGT1A6 (glucuronidation of p-nitrophenol) and UGT2B7 (glucuronidation of androsterone). We showed the inhibitory effect of xanthene dye on human UGT1A6 activity. Basic ET, PL, and RB in those food dyes strongly inhibited UGT1A6 activity, with IC(50) values = 0.05, 0.04, and 0.015 mM, respectively. Meanwhile, AR of an acidic xanthene food dye showed no inhibition. Next, we studied the inhibition of CYP3A4 of a major phase I drug-metabolizing enzyme and P-glycoprotein of a major transporter by synthetic food dyes. Human CYP3A4 and P-glycoprotein were also inhibited by basic xanthene food dyes. The IC(50) values of these dyes to inhibit CYP3A4 and P-glycoprotein were the same as the inhibition level of UGT1A6 by three halogenated xanthene food dyes (ET, PL, and RB) described above, except AR, like the results with UGT1A6 and UGT2B7. We also confirmed the noninhibition of CYP3A4 and P-gp by other synthetic food dyes. Part of this inhibition depended upon the reaction of (1)O(2) originating on xanthene dyes by light irradiation, because inhibition was prevented by (1)O(2) quenchers. We studied the influence of superoxide dismutase and catalase on this inhibition by dyes and we found prevention of inhibition by superoxide dismutase but not catalase. This result suggests that superoxide anions, originating on dyes by light irradiation, must attack drug-metabolizing enzymes. It is possible that red cosmetics containing phloxine, erythrosine, or rose bengal react with proteins on skin under lighting and may lead to rough skin.
Figures
Similar articles
-
Influence of synthetic and natural food dyes on activities of CYP2A6, UGT1A6, and UGT2B7.J Toxicol Environ Health A. 2005 Aug 27;68(16):1431-44. doi: 10.1080/15287390590956588. J Toxicol Environ Health A. 2005. PMID: 16009655
-
Inhibition of human CYP3A4, UGT1A6, and P-glycoprotein with halogenated xanthene food dyes and prevention by superoxide dismutase.J Toxicol Environ Health A. 2008;71(19):1307-13. doi: 10.1080/15287390802240751. J Toxicol Environ Health A. 2008. PMID: 18686201
-
Cytotoxicity of food dyes on cultured fetal rat hepatocytes.Toxicol Appl Pharmacol. 1980 Jun 30;54(2):285-92. doi: 10.1016/0041-008x(80)90198-2. Toxicol Appl Pharmacol. 1980. PMID: 6252664 No abstract available.
-
Biochemical processes mediating neurotoxicity induced by synthetic food dyes: A review of current evidence.Chemosphere. 2024 Sep;364:143295. doi: 10.1016/j.chemosphere.2024.143295. Epub 2024 Sep 12. Chemosphere. 2024. PMID: 39260596 Review.
-
A review of the specifications and toxicity of synthetic food colors permitted in Canada.CRC Crit Rev Toxicol. 1979 Jan;6(2):81-133. doi: 10.3109/10408447909113047. CRC Crit Rev Toxicol. 1979. PMID: 367711 Review. No abstract available.
Cited by
-
Green Synthesis of Molecularly Imprinted Polymers for Dispersive Magnetic Solid-Phase Extraction of Erythrosine B Associated with Smartphone Detection in Food Samples.Materials (Basel). 2022 Oct 31;15(21):7653. doi: 10.3390/ma15217653. Materials (Basel). 2022. PMID: 36363243 Free PMC article.
-
U.S. Food and Drug Administration-Certified Food Dyes as Organocatalysts in the Visible Light-Promoted Chlorination of Aromatics and Heteroaromatics.ACS Omega. 2020 Mar 24;5(13):7693-7704. doi: 10.1021/acsomega.0c00631. eCollection 2020 Apr 7. ACS Omega. 2020. PMID: 32280913 Free PMC article.
-
Antibacterial Activity of Pharmaceutical-Grade Rose Bengal: An Application of a Synthetic Dye in Antibacterial Therapies.Molecules. 2022 Jan 5;27(1):322. doi: 10.3390/molecules27010322. Molecules. 2022. PMID: 35011554 Free PMC article.
-
The promiscuous protein binding ability of erythrosine B studied by metachromasy (metachromasia).J Mol Recognit. 2013 Apr;26(4):181-9. doi: 10.1002/jmr.2263. J Mol Recognit. 2013. PMID: 23456742 Free PMC article.
-
In silico and crystallographic studies identify key structural features of biliverdin IXβ reductase inhibitors having nanomolar potency.J Biol Chem. 2018 Apr 13;293(15):5431-5446. doi: 10.1074/jbc.RA118.001803. Epub 2018 Feb 27. J Biol Chem. 2018. PMID: 29487133 Free PMC article.
References
-
- Nelson DR, Kamataki T, Waxman DJ, et al. The P450 superfamily: update on new sequences, gene mapping, accession numbers, early trivial names of enzymes, and nomenclature. DNA and Cell Biology. 1993;12(1):1–51. - PubMed
-
- Omura T, Sato R. The carbon monoxide-binding pigment of liver microsomes. I. Evidence for its hemoprotein nature. The Journal of Biological Chemistry. 1964;239:2370–2378. - PubMed
-
- Mizutani T. PM frequencies of major CYPs in Asians and Caucasians. Drug Metabolism Reviews. 2003;35(2-3):99–106. - PubMed
-
- Guengerich FP. Common and uncommon cytochrome P450 reactions related to metabolism and chemical toxicity. Chemical Research in Toxicology. 2001;14(6):611–650. - PubMed
-
- Kanou M, Saeki K-I, Kato T-A, Takahashi K, Mizutani T. Study of in vitro glucuronidation of hydroxyquinolines with bovine liver microsomes. Fundamental and Clinical Pharmacology. 2002;16(6):513–517. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

