Critical role of arcuate Y4 receptors and the melanocortin system in pancreatic polypeptide-induced reduction in food intake in mice
- PMID: 20041129
- PMCID: PMC2796177
- DOI: 10.1371/journal.pone.0008488
Critical role of arcuate Y4 receptors and the melanocortin system in pancreatic polypeptide-induced reduction in food intake in mice
Abstract
Background: Pancreatic polypeptide (PP) is a potent anti-obesity agent known to inhibit food intake in the absence of nausea, but the mechanism behind this process is unknown.
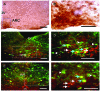
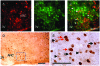
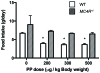
Methodology/principal findings: Here we demonstrate that in response to i.p. injection of PP in wild type but not in Y4 receptor knockout mice, immunostaining for the neuronal activation marker c-Fos is induced specifically in neurons of the nucleus tractus solitarius and the area postrema in the brainstem, notably in cells also showing immunostaining for tyrosine hydroxylase. Importantly, strong c-Fos activation is also detected in the arcuate nucleus of the hypothalamus (ARC), particularly in neurons that co-express alpha melanocyte stimulating hormone (alpha-MSH), the anorexigenic product of the proopiomelanocortin (POMC) gene. Interestingly, other hypothalamic regions such as the paraventricular nucleus, the ventromedial nucleus and the lateral hypothalamic area also show c-Fos induction after PP injection. In addition to c-Fos activation, PP injection up-regulates POMC mRNA expression in the ARC as detected by in situ hybridization. These effects are a direct consequence of local Y4 signaling, since hypothalamus-specific conditional Y4 receptor knockout abolishes PP-induced ARC c-Fos activation and blocks the PP-induced increase in POMC mRNA expression. Additionally, the hypophagic effect of i.p. PP seen in wild type mice is completely absent in melanocortin 4 receptor knockout mice.
Conclusions/significance: Taken together, these findings show that PP reduces food intake predominantly via stimulation of the anorexigenic alpha-MSH signaling pathway, and that this effect is mediated by direct action on local Y4 receptors within the ARC, highlighting a potential novel avenue for the treatment of obesity.
Conflict of interest statement
Figures



Similar articles
-
Pituitary adenylate cyclase-activating polypeptide inhibits food intake in mice through activation of the hypothalamic melanocortin system.Neuropsychopharmacology. 2009 Jan;34(2):424-35. doi: 10.1038/npp.2008.73. Epub 2008 Jun 4. Neuropsychopharmacology. 2009. PMID: 18536705
-
PYY3-36 and pancreatic polypeptide reduce food intake in an additive manner via distinct hypothalamic dependent pathways in mice.Obesity (Silver Spring). 2013 Dec;21(12):E669-78. doi: 10.1002/oby.20534. Epub 2013 Sep 5. Obesity (Silver Spring). 2013. PMID: 23804428
-
Y4 receptors and pancreatic polypeptide regulate food intake via hypothalamic orexin and brain-derived neurotropic factor dependent pathways.Neuropeptides. 2010 Jun;44(3):261-8. doi: 10.1016/j.npep.2010.01.001. Epub 2010 Feb 8. Neuropeptides. 2010. PMID: 20116098
-
The melanocortin pathway and control of appetite-progress and therapeutic implications.J Endocrinol. 2019 Apr 1;241(1):R1-R33. doi: 10.1530/JOE-18-0596. J Endocrinol. 2019. PMID: 30812013 Free PMC article. Review.
-
The leptin-dependent and -independent melanocortin signaling system: regulation of feeding and energy expenditure.J Endocrinol. 2007 Apr;193(1):1-9. doi: 10.1677/JOE-06-0144. J Endocrinol. 2007. PMID: 17400797 Review.
Cited by
-
Cold exposure promotes obesity and impairs glucose homeostasis in mice subjected to a high‑fat diet.Mol Med Rep. 2018 Oct;18(4):3923-3931. doi: 10.3892/mmr.2018.9382. Epub 2018 Aug 10. Mol Med Rep. 2018. PMID: 30106124 Free PMC article.
-
Effect of gut hormones on bone metabolism and their possible mechanisms in the treatment of osteoporosis.Front Pharmacol. 2024 Apr 25;15:1372399. doi: 10.3389/fphar.2024.1372399. eCollection 2024. Front Pharmacol. 2024. PMID: 38725663 Free PMC article. Review.
-
The future role of gut hormones in the treatment of obesity.Ther Adv Chronic Dis. 2014 Jan;5(1):4-14. doi: 10.1177/2040622313506730. Ther Adv Chronic Dis. 2014. PMID: 24381724 Free PMC article. Review.
-
Appetite regulation and weight control: the role of gut hormones.Nutr Diabetes. 2012 Jan 16;2(1):e26. doi: 10.1038/nutd.2011.21. Nutr Diabetes. 2012. PMID: 23154682 Free PMC article.
-
Pancreatic Ppy-expressing γ-cells display mixed phenotypic traits and the adaptive plasticity to engage insulin production.Nat Commun. 2021 Jul 22;12(1):4458. doi: 10.1038/s41467-021-24788-0. Nat Commun. 2021. PMID: 34294685 Free PMC article.
References
-
- (1998) Clinical Guidelines on the Identification, Evaluation, and Treatment of Overweight and Obesity in Adults–The Evidence Report. National Institutes of Health. Obes Res. 6(Suppl 2):51S–209S. - PubMed
-
- Wing RR, Hill JO. Successful weight loss maintenance. Annual Review of Nutrition. 2001;21:323–341. - PubMed
-
- Iyengar S, Li DL, Simmons RM. Characterization of neuropeptide Y-induced feeding in mice: do Y1-Y6 receptor subtypes mediate feeding? Journal of Pharmacology & Experimental Therapeutics. 1999;289:1031–1040. - PubMed
-
- Pedrazzini T, Seydoux J, Kunstner P, Aubert JF, Grouzmann E, et al. Cardiovascular response, feeding behavior and locomotor activity in mice lacking the NPY Y1 receptor. Nature Medicine. 1998;4:722–726. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

