Toxoplasma gondii actin depolymerizing factor acts primarily to sequester G-actin
- PMID: 20042603
- PMCID: PMC2825477
- DOI: 10.1074/jbc.M109.068155
Toxoplasma gondii actin depolymerizing factor acts primarily to sequester G-actin
Abstract
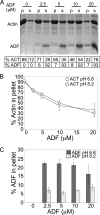
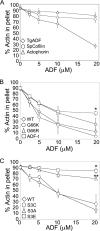
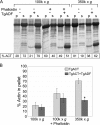
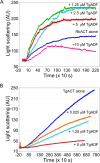
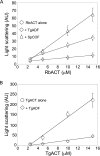
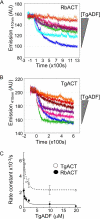
Toxoplasma gondii is a protozoan parasite belonging to the phylum Apicomplexa. Parasites in this phylum utilize a unique process of motility termed gliding, which is dependent on parasite actin filaments. Surprisingly, 98% of parasite actin is maintained as G-actin, suggesting that filaments are rapidly assembled and turned over. Little is known about the regulated disassembly of filaments in the Apicomplexa. In higher eukaryotes, the related actin depolymerizing factor (ADF) and cofilin proteins are essential regulators of actin filament turnover. ADF is one of the few actin-binding proteins conserved in apicomplexan parasites. In this study we examined the mechanism by which T. gondii ADF (TgADF) regulates actin filament turnover. Unlike other members of the ADF/cofilin (AC) family, apicomplexan ADFs lack key F-actin binding sites. Surprisingly, this promotes their enhanced disassembly of actin filaments. Restoration of the C-terminal F-actin binding site to TgADF stabilized its interaction with filaments but reduced its net filament disassembly activity. Analysis of severing activity revealed that TgADF is a weak severing protein, requiring much higher concentrations than typical AC proteins. Investigation of TgADF interaction with T. gondii actin (TgACT) revealed that TgADF disassembled short TgACT oligomers. Kinetic and steady-state polymerization assays demonstrated that TgADF has strong monomer-sequestering activity, inhibiting TgACT polymerization at very low concentrations. Collectively these data indicate that TgADF promoted the efficient turnover of actin filaments via weak severing of filaments and strong sequestering of monomers. This suggests a dual role for TgADF in maintaining high G-actin concentrations and effecting rapid filament turnover.
Figures


References
-
- Joynson D. H., Wreghitt T. J. (2001) Toxoplasmosis: A Comprehensive Clinical Guide, Cambridge University Press, New York
-
- Barragan A., Sibley L. D. (2003) Trends Microbiol. 11, 426–430 - PubMed
-
- Sibley L. D. (2004) Science 304, 248–253 - PubMed
-
- Dobrowolski J. M., Sibley L. D. (1996) Cell 84, 933–939 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

