ALK5 phosphorylation of the endoglin cytoplasmic domain regulates Smad1/5/8 signaling and endothelial cell migration
- PMID: 20042635
- PMCID: PMC2832549
- DOI: 10.1093/carcin/bgp327
ALK5 phosphorylation of the endoglin cytoplasmic domain regulates Smad1/5/8 signaling and endothelial cell migration
Abstract
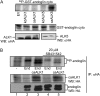
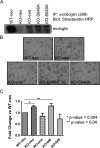
Endoglin, an endothelial cell-specific transforming growth factor-beta (TGF-beta) superfamily coreceptor, has an essential role in angiogenesis. Endoglin-null mice have an embryonic lethal phenotype due to defects in angiogenesis and mutations in endoglin result in the vascular disease hereditary hemorrhagic telangiectasia type I. Increased endoglin expression in the proliferating endothelium of tumors has been correlated with metastasis, tumor grade and decreased survival. Although endoglin is thought to regulate TGF-beta superfamily signaling in endothelial cells through regulating the balance between two TGF-beta-responsive pathways, the activin receptor-like kinase 5 (ALK5)/Smad2/3 pathway and the activin receptor-like kinase 1 (ALK1)/Smad1/5/8 pathway, the mechanism by which endoglin regulates angiogenesis has not been defined. Here, we investigate the role of the cytoplasmic domain of endoglin and its phosphorylation by ALK5 in regulating endoglin function in endothelial cells. We demonstrate that the cytoplasmic domain of endoglin is basally phosphorylated by ALK5, primarily on serines 646 and 649, in endothelial cells. Functionally, the loss of phosphorylation at serine 646 resulted in a loss of endoglin-mediated inhibition of Smad1/5/8 signaling in response to TGF-beta and endothelial cell migration, whereas loss of phosphorylation at both serines 646 and 649 resulted in a loss of endoglin-mediated inhibition of Smad1/5/8 signaling in response to bone morphogenetic protein-9. Taken together, these results support endoglin phosphorylation by ALK5 as an important mechanism for regulating TGF-beta superfamily signaling and migration in endothelial cells.
Figures



Similar articles
-
ALK1 regulates the internalization of endoglin and the type III TGF-β receptor.Mol Biol Cell. 2021 Apr 1;32(7):605-621. doi: 10.1091/mbc.E20-03-0199. Epub 2021 Feb 10. Mol Biol Cell. 2021. PMID: 33566682 Free PMC article.
-
Endoglin promotes transforming growth factor beta-mediated Smad 1/5/8 signaling and inhibits endothelial cell migration through its association with GIPC.J Biol Chem. 2008 Nov 21;283(47):32527-33. doi: 10.1074/jbc.M803059200. Epub 2008 Sep 5. J Biol Chem. 2008. PMID: 18775991 Free PMC article.
-
Endoglin differentially modulates antagonistic transforming growth factor-beta1 and BMP-7 signaling.J Biol Chem. 2007 May 11;282(19):13934-43. doi: 10.1074/jbc.M611062200. Epub 2007 Mar 21. J Biol Chem. 2007. PMID: 17376778
-
Hereditary hemorrhagic telangiectasia, a vascular dysplasia affecting the TGF-beta signaling pathway.Clin Med Res. 2006 Mar;4(1):66-78. doi: 10.3121/cmr.4.1.66. Clin Med Res. 2006. PMID: 16595794 Free PMC article. Review.
-
Controlling the angiogenic switch: a balance between two distinct TGF-b receptor signaling pathways.Trends Cardiovasc Med. 2003 Oct;13(7):301-7. doi: 10.1016/s1050-1738(03)00142-7. Trends Cardiovasc Med. 2003. PMID: 14522471 Review.
Cited by
-
Endoglin regulates the activation and quiescence of endothelium by participating in canonical and non-canonical TGF-β signaling pathways.J Cell Sci. 2013 Mar 15;126(Pt 6):1392-405. doi: 10.1242/jcs.117275. Epub 2013 Feb 15. J Cell Sci. 2013. PMID: 23418351 Free PMC article.
-
Radial Glia Cells Control Angiogenesis in the Developing Cerebral Cortex Through TGF-β1 Signaling.Mol Neurobiol. 2018 May;55(5):3660-3675. doi: 10.1007/s12035-017-0557-8. Epub 2017 May 18. Mol Neurobiol. 2018. PMID: 28523566
-
The Role of Endoglin in Hepatocellular Carcinoma.Int J Mol Sci. 2021 Mar 22;22(6):3208. doi: 10.3390/ijms22063208. Int J Mol Sci. 2021. PMID: 33809908 Free PMC article. Review.
-
The role of endoglin and its soluble form in pathogenesis of preeclampsia.Mol Cell Biochem. 2022 Feb;477(2):479-491. doi: 10.1007/s11010-021-04294-z. Epub 2021 Nov 16. Mol Cell Biochem. 2022. PMID: 34783962 Review.
-
Activins and Inhibins in Cardiovascular Pathophysiology.Biomolecules. 2024 Nov 18;14(11):1462. doi: 10.3390/biom14111462. Biomolecules. 2024. PMID: 39595638 Free PMC article. Review.
References
-
- Pepper MS. Transforming growth factor-beta: vasculogenesis, angiogenesis, and vessel wall integrity. Cytokine Growth Factor Rev. 1997;8:21–43. - PubMed
-
- Attisano L, et al. Identification of human activin and TGF beta type I receptors that form heteromeric kinase complexes with type II receptors. Cell. 1993;75:671–680. - PubMed
-
- Lux A, et al. Assignment of transforming growth factor beta1 and beta3 and a third new ligand to the type I receptor ALK-1. J. Biol. Chem. 1999;274:9984–9992. - PubMed
-
- Goumans MJ, et al. Activin receptor-like kinase (ALK)1 is an antagonistic mediator of lateral TGFbeta/ALK5 signaling. Mol. Cell. 2003;12:817–828. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

