Induction of tolerance to bone marrow allografts by donor-derived host nonreactive ex vivo-induced central memory CD8 T cells
- PMID: 20042725
- PMCID: PMC2837324
- DOI: 10.1182/blood-2009-10-248716
Induction of tolerance to bone marrow allografts by donor-derived host nonreactive ex vivo-induced central memory CD8 T cells
Abstract
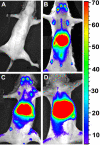
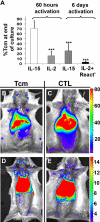
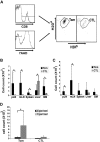
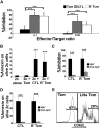
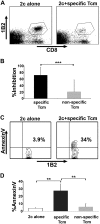
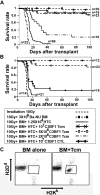
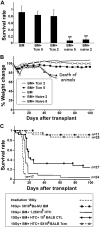
Enabling engraftment of allogeneic T cell-depleted bone marrow (TDBM) under reduced-intensity conditioning represents a major challenge in bone marrow transplantation (BMT). Anti-third-party cytotoxic T lymphocytes (CTLs) were previously shown to be endowed with marked ability to delete host antidonor T cells in vitro, but were found to be less effective in vivo. This could result from diminished lymph node (LN) homing caused by the prolonged activation, which induces a CD44(+)CD62L(-) effector phenotype, and thereby prevents effective colocalization with, and neutralization of, alloreactive host T cells (HTCs). In the present study, LN homing, determined by imaging, was enhanced upon culture conditions that favor the acquisition of CD44(+)CD62L(+) central memory cell (Tcm) phenotype by anti-third-party CD8(+) cells. These Tcm-like cells displayed strong proliferation and prolonged persistence in BM transplant recipients. Importantly, adoptively transferred HTCs bearing a transgenic T-cell receptor (TCR) with antidonor specificity were efficiently deleted only by donor-type Tcms. All these attributes were found to be associated with improved efficacy in overcoming T cell-mediated rejection of TDBM, thereby enabling high survival rate and long-term donor chimerism, without causing graft-versus-host disease. In conclusion, anti-third-party Tcms, which home to recipient LNs and effectively delete antidonor T cells, could provide an effective and novel tool for overcoming rejection of BM allografts.
Figures
Similar articles
-
Murine anti-third-party central-memory CD8(+) T cells promote hematopoietic chimerism under mild conditioning: lymph-node sequestration and deletion of anti-donor T cells.Blood. 2013 Feb 14;121(7):1220-8. doi: 10.1182/blood-2012-07-441493. Epub 2012 Dec 5. Blood. 2013. PMID: 23223359 Free PMC article.
-
Induction of transplantation tolerance in haploidenical transplantation under reduced intensity conditioning: the role of ex-vivo generated donor CD8+ T cells with central memory phenotype.Best Pract Res Clin Haematol. 2011 Sep;24(3):393-401. doi: 10.1016/j.beha.2011.05.007. Epub 2011 Jul 13. Best Pract Res Clin Haematol. 2011. PMID: 21925092 Review.
-
A new approach for eradication of residual lymphoma cells by host nonreactive anti-third-party central memory CD8 T cells.Blood. 2013 Apr 11;121(15):3033-40. doi: 10.1182/blood-2012-06-432443. Epub 2013 Feb 27. Blood. 2013. PMID: 23446736 Free PMC article.
-
Influence of the additional injection of host-type bone marrow on the immune tolerance of minor antigen-mismatched chimeras: possible involvement of double-negative (natural killer) T cells.Transplantation. 1999 Nov 27;68(10):1560-7. doi: 10.1097/00007890-199911270-00021. Transplantation. 1999. PMID: 10589955
-
Mixed chimerism through donor bone marrow transplantation: a tolerogenic cell therapy for application in organ transplantation.Curr Opin Organ Transplant. 2012 Feb;17(1):63-70. doi: 10.1097/MOT.0b013e32834ee68b. Curr Opin Organ Transplant. 2012. PMID: 22186093 Review.
Cited by
-
A strategy to protect off-the-shelf cell therapy products using virus-specific T-cells engineered to eliminate alloreactive T-cells.J Transl Med. 2019 Jul 24;17(1):240. doi: 10.1186/s12967-019-1988-y. J Transl Med. 2019. PMID: 31340822 Free PMC article.
-
Haploidentical SCT: the mechanisms underlying the crossing of HLA barriers.Bone Marrow Transplant. 2014 Jul;49(7):873-9. doi: 10.1038/bmt.2014.19. Epub 2014 Feb 24. Bone Marrow Transplant. 2014. PMID: 24566712 Review.
-
The role of donor-derived veto cells in nonmyeloablative haploidentical HSCT.Bone Marrow Transplant. 2015 Jun;50 Suppl 2:S14-20. doi: 10.1038/bmt.2015.89. Bone Marrow Transplant. 2015. PMID: 26039201
-
5-azacytidine promotes an inhibitory T-cell phenotype and impairs immune mediated antileukemic activity.Mediators Inflamm. 2014;2014:418292. doi: 10.1155/2014/418292. Epub 2014 Mar 13. Mediators Inflamm. 2014. PMID: 24757283 Free PMC article.
-
Anti-viral CD8 central memory veto cells as a new platform for CAR T cell therapy.Stem Cells Transl Med. 2025 May 31;14(6):szaf020. doi: 10.1093/stcltm/szaf020. Stem Cells Transl Med. 2025. PMID: 40448965 Free PMC article.
References
-
- Reisner Y, Kapoor N, Kirkpatrick D, et al. Transplantation for severe combined immunodeficiency with HLA-A,B,D,DR incompatible parental marrow cells fractionated by soybean agglutinin and sheep red blood cells. Blood. 1983;61(2):341–348. - PubMed
-
- Kernan NA, Flomenberg N, Dupont B, O'Reilly RJ. Graft rejection in recipients of T-cell-depleted HLA-nonidentical marrow transplants for leukemia: identification of host-derived antidonor allocytotoxic T lymphocytes. Transplantation. 1987;43(6):842–847. - PubMed
-
- Gale RP, Reisner Y. Graft rejection and graft-versus-host disease: mirror images. Lancet. 1986;1(8496):1468–1470. - PubMed
-
- Terenzi A, Aversa F, Albi N, et al. Residual clonable host cell detection for predicting engraftment of T cell depleted BMTs. Bone Marrow Transplant. 1993;11(5):357–361. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

