Acetylsalicylic acid enhances purinergic receptor-mediated outward currents in rat megakaryocytes
- PMID: 20042731
- PMCID: PMC2838576
- DOI: 10.1152/ajpcell.00422.2009
Acetylsalicylic acid enhances purinergic receptor-mediated outward currents in rat megakaryocytes
Abstract
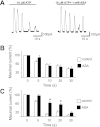
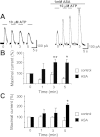
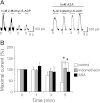
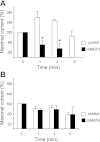
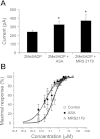
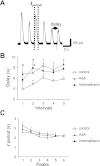
Purinergic receptor activation increases cytosolic Ca(2+) concentration in a fluctuating fashion, triggering oscillatory outward Ca(2+)-activated K(+) currents in rat megakaryocytes (MKs). Whole cell and nystatin-perforated patch-clamp techniques were used to analyze changes in ionic conductance in MK with acetylsalicylic acid (ASA), a cyclooxygenase-1 inhibitor and antithrombotic agent. MKs are a model for platelet reactivity, particularly in ASA treatment failure (ASA resistance). Freshly isolated MKs were incubated 30 min in the absence or presence of 1 mM ASA. Using a K(+)-rich internal solution, we recorded outward currents in response to 10 microM ATP, 10 microM ADP, and 5 microM 2-methyl-thio-ADP (2MeSADP) in the voltage-clamp mode. Agonist-induced currents decreased in amplitude over time, but this decline was attenuated by ASA in both continuous and repeated agonist challenge, indicating increased MK reactivity with ASA treatment. In separate experiments, heterologous desensitization was observed when MKs were stimulated with ADP after exposure to a thromboxane receptor agonist (U46619), indicating cross talk between thromboxane and purinergic pathways. Different cells, treated with ASA or MRS2179 (P2Y1 receptor antagonist), were stimulated with 2MeSADP. The dose-response curve was shifted to the left in both cases, suggesting increased MK reactivity. ASA also caused an increased interval between currents (delay). ASA attenuated desensitization of purinergic receptors and increased delay, again suggesting cross talk between purinergic and thromboxane pathways. These findings may be relevant to ASA resistance, because individual variations in sensitivity to the multiple effects of ASA on signaling pathways could result in insensitivity to its antiplatelet effects in some patients.
Figures


References
-
- A randomised, blinded, trial of clopidogrel versus aspirin in patients at risk of ischaemic events (CAPRIE) CAPRIE Steering Committee. Lancet 348: 1329–1339, 1996 - PubMed
-
- Authi KS. TRP channels in platelet function. Handb Exp Pharmacol 179: 425–443, 2007 - PubMed
-
- Barton JF, Hardy AR, Poole AW, Mundell SJ. Reciprocal regulation of platelet responses to P2Y and thromboxane receptor activation. J Thromb Haemost 6: 534–543, 2008 - PubMed
-
- Baurand A, Eckly A, Hechler A, Kauffenstein G, Galzi JL, Cazenave JP, Léon C, Gachet C. Differential regulation and relocalization of the platelet P2Y receptors after activation: a way to avoid loss of hemostatic properties? Mol Pharmacol 67: 721–733, 2005 - PubMed
-
- Bergmeier W, Stefanini L. Novel molecules in calcium signaling in platelets. J Thromb Haemost 7, Suppl 1: 187–190, 2009 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

