Monoketone analogs of curcumin, a new class of Fanconi anemia pathway inhibitors
- PMID: 20043851
- PMCID: PMC2807854
- DOI: 10.1186/1476-4598-8-133
Monoketone analogs of curcumin, a new class of Fanconi anemia pathway inhibitors
Abstract
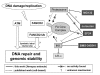
Background: The Fanconi anemia (FA) pathway is a multigene DNA damage response network implicated in the repair of DNA lesions that arise during replication or after exogenous DNA damage. The FA pathway displays synthetic lethal relationship with certain DNA repair genes such as ATM (Ataxia Telangectasia Mutated) that are frequently mutated in tumors. Thus, inhibition of FANCD2 monoubiquitylation (FANCD2-Ub), a key step in the FA pathway, might target tumor cells defective in ATM through synthetic lethal interaction. Curcumin was previously identified as a weak inhibitor of FANCD2-Ub. The aim of this study is to identify derivatives of curcumin with better activity and specificity.
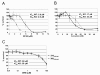
Results: Using a replication-free assay in Xenopus extracts, we screened monoketone analogs of curcumin for inhibition of FANCD2-Ub and identified analog EF24 as a strong inhibitor. Mechanistic studies suggest that EF24 targets the FA pathway through inhibition of the NF-kB pathway kinase IKK. In HeLa cells, nanomolar concentrations of EF24 inhibited hydroxyurea (HU)-induced FANCD2-Ub and foci in a cell-cycle independent manner. Survival assays revealed that EF24 specifically sensitizes FA-competent cells to the DNA crosslinking agent mitomycin C (MMC). In addition, in contrast with curcumin, ATM-deficient cells are twofold more sensitive to EF24 than matched wild-type cells, consistent with a synthetic lethal effect between FA pathway inhibition and ATM deficiency. An independent screen identified 4H-TTD, a compound structurally related to EF24 that displays similar activity in egg extracts and in cells.
Conclusions: These results suggest that monoketone analogs of curcumin are potent inhibitors of the FA pathway and constitute a promising new class of targeted anticancer compounds.
Figures

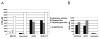



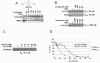
Similar articles
-
Proteasome function is required for DNA damage response and fanconi anemia pathway activation.Cancer Res. 2007 Aug 1;67(15):7395-405. doi: 10.1158/0008-5472.CAN-07-1015. Cancer Res. 2007. PMID: 17671210
-
Fanconi anemia pathway-deficient tumor cells are hypersensitive to inhibition of ataxia telangiectasia mutated.J Clin Invest. 2007 May;117(5):1440-9. doi: 10.1172/JCI31245. Epub 2007 Apr 12. J Clin Invest. 2007. PMID: 17431503 Free PMC article.
-
Convergence of the fanconi anemia and ataxia telangiectasia signaling pathways.Cell. 2002 May 17;109(4):459-72. doi: 10.1016/s0092-8674(02)00747-x. Cell. 2002. PMID: 12086603
-
The Fanconi anemia ID2 complex: dueling saxes at the crossroads.Cell Cycle. 2014;13(19):2999-3015. doi: 10.4161/15384101.2014.956475. Cell Cycle. 2014. PMID: 25486561 Free PMC article. Review.
-
The interplay of Fanconi anemia proteins in the DNA damage response.DNA Repair (Amst). 2004 Aug-Sep;3(8-9):1063-9. doi: 10.1016/j.dnarep.2004.04.005. DNA Repair (Amst). 2004. PMID: 15279794 Review.
Cited by
-
Monocarbonyl curcumin analogues: heterocyclic pleiotropic kinase inhibitors that mediate anticancer properties.J Med Chem. 2013 May 9;56(9):3456-66. doi: 10.1021/jm4002692. Epub 2013 Apr 23. J Med Chem. 2013. PMID: 23550937 Free PMC article.
-
Selective tumor killing based on specific DNA-damage response deficiencies.Cancer Biol Ther. 2012 Mar;13(5):239-46. doi: 10.4161/cbt.18921. Epub 2012 Mar 1. Cancer Biol Ther. 2012. PMID: 22258411 Free PMC article. Review.
-
Targeting the fanconi anemia pathway to identify tailored anticancer therapeutics.Anemia. 2012;2012:481583. doi: 10.1155/2012/481583. Epub 2012 May 24. Anemia. 2012. PMID: 22693661 Free PMC article.
-
Monocarbonyl analogs of curcumin inhibit growth of antibiotic sensitive and resistant strains of Mycobacterium tuberculosis.Eur J Med Chem. 2015 Mar 6;92:693-9. doi: 10.1016/j.ejmech.2015.01.020. Epub 2015 Jan 10. Eur J Med Chem. 2015. PMID: 25618016 Free PMC article.
-
CLEFMA-an anti-proliferative curcuminoid from structure-activity relationship studies on 3,5-bis(benzylidene)-4-piperidones.Bioorg Med Chem. 2010 Aug 15;18(16):6109-20. doi: 10.1016/j.bmc.2010.06.055. Epub 2010 Jun 22. Bioorg Med Chem. 2010. PMID: 20638855 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

