The effect of 17 beta-estradiol on intracellular calcium homeostasis in human endothelial cells
- PMID: 20044991
- PMCID: PMC2822064
- DOI: 10.1016/j.ejphar.2009.12.030
The effect of 17 beta-estradiol on intracellular calcium homeostasis in human endothelial cells
Abstract
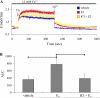
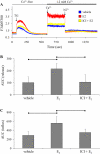
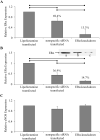
The cardiovascular effects of estrogen are mediated in part by augmenting the function of endothelial nitric oxide synthase. Endothelial nitric oxide synthase activity is dependent on many cofactors including Ca(2+). Hence, we investigated the effect of chronic 17 beta-estradiol treatment on the intracellular Ca(2+) concentration and endothelial nitric oxide synthase protein expression in the human endothelial cell line, EA.hy926, using spectrofluorometry and Western blot, respectively. Inhibiting the sarco(endo)plasmic reticulum Ca(2+) ATPase with thapsigargin caused an increase in the intracellular Ca(2+) concentration, which was higher in chronically 17 beta-estradiol-treated (1muM, 24h) cells loaded with Fura-2-acetoxymethyl ester compared to vehicle-treated cells, suggesting a higher endoplasmic reticulum Ca(2+) content in 17 beta-estradiol-treated cells. An enhanced Ca(2+) influx pathway in chronically 17 beta-estradiol-treated cells was also observed. In addition, 17 beta-estradiol-treated cells expressed higher levels of endothelial nitric oxide synthase protein in comparison to vehicle-treated cells. The chronic effect of 17 beta-estradiol on Ca(2+) homeostasis and endothelial nitric oxide synthase expression was attenuated with the nonselective estrogen receptor inhibitor, ICI 182,780 (10muM, 7alpha, 17beta-[9-[(4,4,5,5,5-Pentafluoropentyl)sulfinyl]nonyl] estra-1,3,5(10)-triene-3,17-diol). Furthermore, analysis of the thapsigargin-evoked Ca(2+) response in chronically 17 beta-estradiol-treated estrogen receptor alpha-knockdown cells showed no significant difference in Ca(2+) response compared to vehicle-treated estrogen receptor alpha-knockdown cells, indicating that the regulation of Ca(2+) homeostasis by 17 beta-estradiol is mediated through an estrogen receptor alpha-dependent pathway. These data revealed an estrogen receptor alpha-dependent modulation of Ca(2+) homeostasis accompanying the enhancement of endothelial nitric oxide synthase expression in 17 beta-estradiol-treated human endothelial cells.
Figures
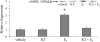

Similar articles
-
Effects of 17 β-estradiol on lipopolysacharride-induced intracellular adhesion molecule-1 mRNA expression and Ca²+ homeostasis alteration in human endothelial cells.Vascul Pharmacol. 2010 Nov-Dec;53(5-6):230-8. doi: 10.1016/j.vph.2010.09.001. Epub 2010 Sep 16. Vascul Pharmacol. 2010. PMID: 20843480 Free PMC article.
-
Estrogen increases nitric-oxide production in human bronchial epithelium.J Pharmacol Exp Ther. 2011 Dec;339(3):815-24. doi: 10.1124/jpet.111.184416. Epub 2011 Sep 22. J Pharmacol Exp Ther. 2011. PMID: 21940647 Free PMC article.
-
Soy isoflavones improve endothelial function in spontaneously hypertensive rats in an estrogen-independent manner: role of nitric-oxide synthase, superoxide, and cyclooxygenase metabolites.J Pharmacol Exp Ther. 2005 Sep;314(3):1300-9. doi: 10.1124/jpet.105.085530. Epub 2005 Jun 15. J Pharmacol Exp Ther. 2005. PMID: 15958720
-
Regulation of endothelial nitric oxide synthase and high-density lipoprotein quality by estradiol in cardiovascular pathology.J Cardiovasc Pharmacol Ther. 2014 May;19(3):256-68. doi: 10.1177/1074248413513499. Epub 2014 Jan 12. J Cardiovasc Pharmacol Ther. 2014. PMID: 24414281 Review.
-
Rapid activation of endothelial NO synthase by estrogen: evidence for a steroid receptor fast-action complex (SRFC) in caveolae.Steroids. 2002 May;67(6):413-9. doi: 10.1016/s0039-128x(01)00177-5. Steroids. 2002. PMID: 11960616 Review.
Cited by
-
SGK3 sustains ERα signaling and drives acquired aromatase inhibitor resistance through maintaining endoplasmic reticulum homeostasis.Proc Natl Acad Sci U S A. 2017 Feb 21;114(8):E1500-E1508. doi: 10.1073/pnas.1612991114. Epub 2017 Feb 7. Proc Natl Acad Sci U S A. 2017. PMID: 28174265 Free PMC article.
-
Sexual Dimorphism in Migraine. Focus on Mitochondria.Curr Pain Headache Rep. 2025 Jan 6;29(1):11. doi: 10.1007/s11916-024-01317-4. Curr Pain Headache Rep. 2025. PMID: 39760955 Review.
-
17β-Estradiol promotes sex-specific dysfunction in isolated human arterioles.Am J Physiol Heart Circ Physiol. 2023 Mar 1;324(3):H330-H337. doi: 10.1152/ajpheart.00708.2022. Epub 2023 Jan 6. Am J Physiol Heart Circ Physiol. 2023. PMID: 36607795 Free PMC article.
-
Association of reproductive factors with dementia: A systematic review and dose-response meta-analyses of observational studies.EClinicalMedicine. 2021 Dec 14;43:101236. doi: 10.1016/j.eclinm.2021.101236. eCollection 2022 Jan. EClinicalMedicine. 2021. PMID: 34977513 Free PMC article.
-
17β-Estradiol Increases APE1/Ref-1 Secretion in Vascular Endothelial Cells and Ovariectomized Mice: Involvement of Calcium-Dependent Exosome Pathway.Biomedicines. 2021 Aug 18;9(8):1040. doi: 10.3390/biomedicines9081040. Biomedicines. 2021. PMID: 34440244 Free PMC article.
References
-
- Barrett-Connor E, Bush TL. Estrogen and coronary heart disease in women. JAMA. 1991;265:1861–1867. - PubMed
-
- Bryan RM, Jr., You J, Golding EM, Marrelli SP. Endothelium-derived hyperpolarizing factor: a cousin to nitric oxide and prostacyclin. Anesthesiology. 2005;102:1261–1277. - PubMed
-
- Busse R, Edwards G, Feletou M, Fleming I, Vanhoutte PM, Weston AH. EDHF: bringing the concepts together. Trends Pharmacol Sci. 2002;23:374–380. - PubMed
-
- Deenadayalu VP, White RE, Stallone JN, Gao X, Garcia AJ. Testosterone relaxes coronary arteries by opening the large-conductance, calcium-activated potassium channel. Am J Physiol Heart Circ Physiol. 2001;281:H1720–1727. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

