Cyclic AMP negatively regulates prodigiosin production by Serratia marcescens
- PMID: 20045458
- PMCID: PMC2846241
- DOI: 10.1016/j.resmic.2009.12.004
Cyclic AMP negatively regulates prodigiosin production by Serratia marcescens
Abstract
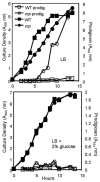
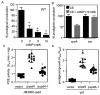
Many Serratia marcescens strains produce the red pigment prodigiosin, which has antimicrobial and anti-tumor properties. Previous reports suggest that cyclic AMP (cAMP) is a positive regulator of prodigiosin production. Supporting this model, the addition of glucose to growth medium inhibited pigment production in rich and minimal media. Unexpectedly, we observed highly elevated levels of prodigiosin production in isogenic strains with mutations in genes involved in cAMP production (cyaA and crr) and in cAMP-dependent transcriptional signaling (crp). Multicopy expression of the Escherichia coli cAMP-phosphodiesterase gene, cpdA, also conferred a striking increase in prodigiosin production. Exogenous cAMP decreased both pigment production and pigA-lacZ transcription in the wild-type (WT) strain, and pigA-lacZ transcription was significantly increased in a crp mutant relative to WT. Suppressor and epistasis analysis indicate that the hyperpigment phenotype was dependent upon pigment biosynthetic genes (pigA, pigB, pigC, pigD and pigM). These experiments establish cAMP as a negative regulator of prodigiosin production in S. marcescens.
Copyright 2009 Elsevier Masson SAS. All rights reserved.
Figures

References
-
- Clements-Jewery S. The reversal of glucose repressed prodigiosin production in Serratia marcescens by the cyclic 3′5′-adenosine monophosphate inhibitor theophylline. Experientia. 1976;32:421–422. - PubMed
-
- Coulthurst SJ, Kurz CL, Salmond GP. luxS mutants of Serratia defective in autoinducer-2-dependent ‘quorum sensing’ show strain-dependent impacts on virulence and production of carbapenem and prodigiosin. Microbiology. 2004;150:1901–1910. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

