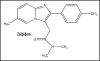
Zolpidem, a clinical hypnotic that affects electronic transfer, alters synaptic activity through potential GABA receptors in the nervous system without significant free radical generation
- PMID: 20046645
- PMCID: PMC2763231
- DOI: 10.4161/oxim.2.1.7859
Zolpidem, a clinical hypnotic that affects electronic transfer, alters synaptic activity through potential GABA receptors in the nervous system without significant free radical generation
Abstract
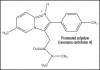
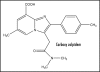
Zolpidem (trade name Ambien) has attracted much interest as a sleep-inducing agent and also in research. Attention has been centered mainly on receptor binding and electrochemistry in the central nervous system which are briefly addressed herein. A novel integrated approach to mode of action is presented. The pathways to be discussed involve basicity, reduction potential, electrostatics, cell signaling, GABA receptor binding, electron transfer (ET), pharmacodynamics, structure activity relationships (SAR) and side effects. The highly conjugated pyridinium salt formed by protonation of the amidine moiety is proposed to be the active form acting as an ET agent. Extrapolation of reduction potentials for related compounds supports the premise that zolpidem may act as an ET species in vivo. From recent literature reports, electrostatics is believed to play a significant role in drug action. The pyridinium cation displays molecular electrostatic potential which may well play a role energetically or as a bridging mechanism. An SAR analysis points to analogy with other physiologically active xenobiotics, namely benzodiazepines and paraquat in the conjugated iminium category. Inactivity of metabolites indicates that the parent is the active form of zolpidem. Absence of reactive oxygen species and oxidative stress is in line with minor side effects. In contrast, generally, the prior literature contains essentially no discussion of these fundamental biochemical relationships. Pharmacodynamics may play an important role. Concerning behavior at the blood-brain barrier, useful insight can be gained from investigations of the related cationic anesthetics that are structurally related to acetyl choline. Evidently, the neutral form of the drug penetrates the neuronal membrane, with the salt form operating at the receptor. The pathways of zolpidem have several clinical implications since the agent affects sedation, electroencephalographic activity, oxidative metabolites and receptors in the central nervous system. The drug acts at the GABA(A) receptor benzodiazepine site, displaying high and intermediate affinities to various receptor regions. Structural features for tight binding were determined. The sedative and anticonvulsant activities are due to its action on the alpha-1-GABA(A) receptors. One of the common adverse responses to zolpidem is hallucinations. Proposed mechanisms comprise changes in the GABA(A) receptor, pharmacodynamic interactions involving serotonin and neuronal-weak photon emission processes entailing redox phenomena. Reports cite cases of abuse with cravings based on anxiolytic and stimulating actions. It is important to recognize that insight concerning processes at the fundamental, molecular level can translate into beneficial results involving both positive and adverse side effects. In order for this to occur, interdisciplinary interaction is necessary. Suggestions are made for future research aimed at testing the various hypotheses.
Keywords: SAR; cell signaling; electrochemistry; neuroscience; receptors; zolpidem.
Figures





Similar articles
-
Mechanism of action of the hypnotic zolpidem in vivo.Br J Pharmacol. 2000 Dec;131(7):1251-4. doi: 10.1038/sj.bjp.0703717. Br J Pharmacol. 2000. PMID: 11090095 Free PMC article.
-
Dissociating anxiolytic and sedative effects of GABAAergic drugs using temperature and locomotor responses to acute stress.Psychopharmacology (Berl). 2009 Jun;204(2):299-311. doi: 10.1007/s00213-009-1460-4. Epub 2009 Jan 24. Psychopharmacology (Berl). 2009. PMID: 19169673 Free PMC article.
-
Zolpidem, a novel nonbenzodiazepine hypnotic. I. Neuropharmacological and behavioral effects.J Pharmacol Exp Ther. 1986 May;237(2):649-58. J Pharmacol Exp Ther. 1986. PMID: 2871178
-
GABA(A)-receptor subtypes: clinical efficacy and selectivity of benzodiazepine site ligands.Ann Med. 1997 Aug;29(4):275-82. doi: 10.3109/07853899708999348. Ann Med. 1997. PMID: 9375983 Review.
-
Comparative pharmacokinetics and pharmacodynamics of short-acting hypnosedatives: zaleplon, zolpidem and zopiclone.Clin Pharmacokinet. 2004;43(4):227-38. doi: 10.2165/00003088-200443040-00002. Clin Pharmacokinet. 2004. PMID: 15005637 Review.
Cited by
-
Multifaceted approach to resveratrol bioactivity: Focus on antioxidant action, cell signaling and safety.Oxid Med Cell Longev. 2010 Mar-Apr;3(2):86-100. doi: 10.4161/oxim.3.2.11147. Oxid Med Cell Longev. 2010. PMID: 20716933 Free PMC article. Review.
-
Clinical physiology and mechanism of dizocilpine (MK-801): electron transfer, radicals, redox metabolites and bioactivity.Oxid Med Cell Longev. 2010 Jan-Feb;3(1):13-22. doi: 10.4161/oxim.3.1.10028. Oxid Med Cell Longev. 2010. PMID: 20716924 Free PMC article. Review.
-
Pharmacotherapeutic management of insomnia and effects on sleep processes, neural plasticity, and brain systems modulating stress: A narrative review.Front Neurosci. 2022 Jul 29;16:893015. doi: 10.3389/fnins.2022.893015. eCollection 2022. Front Neurosci. 2022. PMID: 35968380 Free PMC article. Review.
-
Zolpidem withdrawal delirium.Indian J Pharmacol. 2011 Nov;43(6):729-30. doi: 10.4103/0253-7613.89838. Indian J Pharmacol. 2011. PMID: 22144786 Free PMC article.
-
Development and Validation of a Stability-Indicating Capillary Electrophoresis Method for the Determination of Zolpidem Tartrate in Tablet Dosage Form with Positive Confirmation using 2D- and 3D-DAD Fingerprints.Sci Pharm. 2014 Feb 27;82(2):341-56. doi: 10.3797/scipharm.1401-11. Print 2014 Apr-Jun. Sci Pharm. 2014. PMID: 24959406 Free PMC article.
References
-
- Kovacic P, Pozos RS. Review: Bioelectronome. Integrated approach to receptor chemistry, radical, electron transfer, electrochemistry, cell signaling and physiological effects based on electron transfer. J Recept Signal Transduct. 2007;27:261–294. - PubMed
-
- Kovacic P, Pozos RS. Cell signaling (mechanism and reproductive toxicity): Redox chains, radicals, electrons, relays, conduit, electrochemistry and other medical implications. Birth Defects Res Part C. 2006;78:333–344. - PubMed
-
- Kovacic P, Becvar LE. Mode of action of anti-infective agents: Emphasis on oxidative stress and electron transfer. Curr Pharmaceut Des. 2000;6:143–167. - PubMed
-
- Kovacic P, Osuna JA. Mechanism of anticancer agents: Emphasis on oxidative stress and electron transfer. Curr Pharmaceut Des. 2000;6:277–309. - PubMed
-
- Kovacic P, Jacintho JD. Mechanism of carcinogenesis: focus on oxidative stress and electron transfer. Curr Med Chem. 2001;8:773–796. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

