DNA-mediated charge transport in redox sensing and signaling
- PMID: 20047321
- PMCID: PMC2902267
- DOI: 10.1021/ja907669c
DNA-mediated charge transport in redox sensing and signaling
Abstract
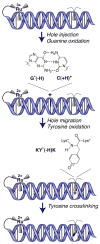
The transport of charge through the DNA base-pair stack offers a route to carry out redox chemistry at a distance. Here we describe characteristics of this chemistry that have been elucidated and how this chemistry may be utilized within the cell. The shallow distance dependence associated with these redox reactions permits DNA-mediated signaling over long molecular distances in the genome and facilitates the activation of redox-sensitive transcription factors globally in response to oxidative stress. The long-range funneling of oxidative damage to sites of low oxidation potential in the genome also may provide a means of protection within the cell. Furthermore, the sensitivity of DNA charge transport to perturbations in base-pair stacking, as may arise with base lesions and mismatches, may be used as a route to scan the genome for damage as a first step in DNA repair. Thus, the ability of double-helical DNA in mediating redox chemistry at a distance provides a natural mechanism for redox sensing and signaling in the genome.
Figures






References
-
- Delaney S, Barton JK. J Org Chem. 2003;68:6475. - PubMed
-
- O'Neill MA, Barton JK. In: Topics in Current Chemistry. Schuster GB, editor. Vol. 236. Springer; Berlin: 2004. p. 67.
-
-
DNA-mediated CT is also facile through DNA/RNA hybrids. See Sartor V, Henderson PT, Schuster GB. J Am Chem Soc. 1999;121:11027.O'Neill MA, Barton JK. J Am Chem Soc. 2002;124:13053.
-
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

