QTLs and candidate genes for desiccation and abscisic acid content in maize kernels
- PMID: 20047666
- PMCID: PMC2826337
- DOI: 10.1186/1471-2229-10-2
QTLs and candidate genes for desiccation and abscisic acid content in maize kernels
Abstract
Background: Kernel moisture at harvest is an important trait since a low value is required to prevent unexpected early germination and ensure seed preservation. It is also well known that early germination occurs in viviparous mutants, which are impaired in abscisic acid (ABA) biosynthesis. To provide some insight into the genetic determinism of kernel desiccation in maize, quantitative trait loci (QTLs) were detected for traits related to kernel moisture and ABA content in both embryo and endosperm during kernel desiccation. In parallel, the expression and mapping of genes involved in kernel desiccation and ABA biosynthesis, were examined to detect candidate genes.
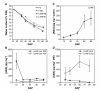
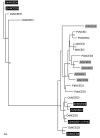
Results: The use of an intermated recombinant inbred line population allowed for precise QTL mapping. For 29 traits examined in an unreplicated time course trial of days after pollination, a total of 78 QTLs were detected, 43 being related to kernel desiccation, 15 to kernel weight and 20 to ABA content. Multi QTL models explained 35 to 50% of the phenotypic variation for traits related to water status, indicating a large genetic control amenable to breeding. Ten of the 20 loci controlling ABA content colocated with previously detected QTLs controlling water status and ABA content in water stressed leaves. Mapping of candidate genes associated with kernel desiccation and ABA biosynthesis revealed several colocations between genes with putative functions and QTLs. Parallel investigation via RT-PCR experiments showed that the expression patterns of the ABA-responsive Rab17 and Rab28 genes as well as the late embryogenesis abundant Emb5 and aquaporin genes were related to desiccation rate and parental allele effect. Database searches led to the identification and mapping of two zeaxanthin epoxidase (ZEP) and five novel 9-cis-epoxycarotenoid dioxygenase (NCED) related genes, both gene families being involved in ABA biosynthesis. The expression of these genes appeared independent in the embryo and endosperm and not correlated with ABA content in either tissue.
Conclusions: A high resolution QTL map for kernel desiccation and ABA content in embryo and endosperm showed several precise colocations between desiccation and ABA traits. Five new members of the maize NCED gene family and another maize ZEP gene were identified and mapped. Among all the identified candidates, aquaporins and members of the Responsive to ABA gene family appeared better candidates than NCEDs and ZEPs.
Figures

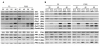
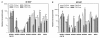
Similar articles
-
QTL mapping and omics analysis to identify genes controlling kernel dehydration in maize.Theor Appl Genet. 2024 Sep 26;137(10):233. doi: 10.1007/s00122-024-04715-9. Theor Appl Genet. 2024. PMID: 39325221
-
Characterization of QTL for oil content in maize kernel.Theor Appl Genet. 2012 Oct;125(6):1169-79. doi: 10.1007/s00122-012-1903-x. Epub 2012 Jun 6. Theor Appl Genet. 2012. PMID: 22669301
-
Abscisic acid biosynthesis in tomato: regulation of zeaxanthin epoxidase and 9-cis-epoxycarotenoid dioxygenase mRNAs by light/dark cycles, water stress and abscisic acid.Plant Mol Biol. 2000 Apr;42(6):833-45. doi: 10.1023/a:1006448428401. Plant Mol Biol. 2000. PMID: 10890531
-
Hotspot Regions of Quantitative Trait Loci and Candidate Genes for Ear-Related Traits in Maize: A Literature Review.Genes (Basel). 2023 Dec 21;15(1):15. doi: 10.3390/genes15010015. Genes (Basel). 2023. PMID: 38275597 Free PMC article. Review.
-
QTL analysis to study the association between leaf size and abscisic acid accumulation in droughted rice leaves and comparisons across cereals.Plant Mol Biol. 1997 Sep;35(1-2):155-65. Plant Mol Biol. 1997. PMID: 9291969 Review.
Cited by
-
The genetic architecture of grain yield and related traits in Zea maize L. revealed by comparing intermated and conventional populations.Genetics. 2010 Sep;186(1):395-404. doi: 10.1534/genetics.110.113878. Epub 2010 Jun 30. Genetics. 2010. PMID: 20592258 Free PMC article.
-
Quantitative trait loci involved in regulating seed oil composition in Arabidopsis thaliana and their evolutionary implications.Theor Appl Genet. 2012 Mar;124(4):723-38. doi: 10.1007/s00122-011-1742-1. Epub 2011 Nov 10. Theor Appl Genet. 2012. PMID: 22072101
-
The ZmASR1 protein influences branched-chain amino acid biosynthesis and maintains kernel yield in maize under water-limited conditions.Plant Physiol. 2011 Oct;157(2):917-36. doi: 10.1104/pp.111.176818. Epub 2011 Aug 18. Plant Physiol. 2011. PMID: 21852416 Free PMC article.
-
Genome-Wide Association Study Reveals the Genetic Basis of Kernel and Cob Moisture Changes in Maize at Physiological Maturity Stage.Plants (Basel). 2022 Jul 30;11(15):1989. doi: 10.3390/plants11151989. Plants (Basel). 2022. PMID: 35956467 Free PMC article.
-
Genetic dissection of grain water content and dehydration rate related to mechanical harvest in maize.BMC Plant Biol. 2020 Mar 17;20(1):118. doi: 10.1186/s12870-020-2302-0. BMC Plant Biol. 2020. PMID: 32183696 Free PMC article.
References
-
- Borras L, Westgate ME. Predicting maize kernel sink capacity early in development. Field Crops Res. 2006;95:223–233. doi: 10.1016/j.fcr.2005.03.001. - DOI
-
- Dwyer LM, Ma BL, Evenson L, Hamilton RI. Maize physiological traits related to grain yield and harvest moisture in mid- to short-season environments. Crop Sci. 1994;34(4):985–992.
-
- Eta-Ndu JT, Openshaw SJ. Selection criteria for grain yield and moisture in maize trials. Crop Sci. 1992;32(2):332–335.
-
- Hawbaker MS, Hill WH, Goodman MM. Application of recurrent selection for low grain moisture content at harvest in tropical maize. Crop Sci. 1997;37(5):1650–1655.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

