Characterization of melanoma cells capable of propagating tumors from a single cell
- PMID: 20048081
- PMCID: PMC4026296
- DOI: 10.1158/0008-5472.CAN-09-2153
Characterization of melanoma cells capable of propagating tumors from a single cell
Abstract
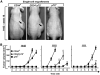
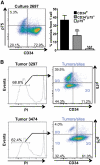
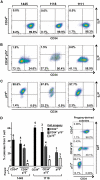
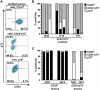
Questions persist about the nature and number of cells with tumor-propagating capability in different types of cancer, including melanoma. In part, this is because identification and characterization of purified tumorigenic subsets of cancer cells has not been achieved to date. Here, we report tumor formation after injection of single purified melanoma cells derived from three novel mouse models. Tumor formation occurred after every injection of individual CD34+p75- melanoma cells, with intermediate rates using CD34-p75- cells, and rarely with CD34-p75+ cells. These findings suggest that tumorigenic melanoma cells may be more common than previously thought and establish that multiple distinct populations of melanoma-propagating cells (MPC) can exist within a single tumor. Interestingly, individual CD34-p75- MPCs could regenerate cellular heterogeneity after tumor formation in mice or multiple passages in vitro, whereas CD34+p75- MPCs underwent self-renewal only, showing that reestablishment of tumor heterogeneity is not always a characteristic of individual cells capable of forming tumors. Functionally, single purified MPCs were more resistant to chemotherapy than non-MPCs. We anticipate that purification of these MPCs may allow a more comprehensive evaluation of the molecular features that define tumor-forming capability and chemotherapeutic resistance in melanoma.
Figures

Similar articles
-
Identification of very small cancer stem cells expressing hallmarks of pluripotency in B16F10 melanoma cells and their reoccurrence in B16F10-derived clones.Exp Cell Res. 2020 Jun 15;391(2):111938. doi: 10.1016/j.yexcr.2020.111938. Epub 2020 Apr 9. Exp Cell Res. 2020. PMID: 32278688
-
BRAF inhibition in melanoma is associated with the dysregulation of histone methylation and histone methyltransferases.Neoplasia. 2020 Sep;22(9):376-389. doi: 10.1016/j.neo.2020.06.006. Epub 2020 Jul 3. Neoplasia. 2020. PMID: 32629178 Free PMC article.
-
Patterns of immune-cell infiltration in murine models of melanoma: roles of antigen and tissue site in creating inflamed tumors.Cancer Immunol Immunother. 2019 Jul;68(7):1121-1132. doi: 10.1007/s00262-019-02345-5. Epub 2019 May 27. Cancer Immunol Immunother. 2019. PMID: 31134297 Free PMC article.
-
Aryl hydrocarbon receptor/cytochrome P450 1A1 pathway mediates breast cancer stem cells expansion through PTEN inhibition and β-Catenin and Akt activation.Mol Cancer. 2017 Jan 19;16(1):14. doi: 10.1186/s12943-016-0570-y. Mol Cancer. 2017. PMID: 28103884 Free PMC article.
-
Human relevance of NRAS/BRAF mouse melanoma models.Eur J Cell Biol. 2014 Jan-Feb;93(1-2):82-6. doi: 10.1016/j.ejcb.2013.10.010. Epub 2013 Nov 4. Eur J Cell Biol. 2014. PMID: 24342721 Review.
Cited by
-
BRAFV600E cooperates with PI3K signaling, independent of AKT, to regulate melanoma cell proliferation.Mol Cancer Res. 2014 Mar;12(3):447-63. doi: 10.1158/1541-7786.MCR-13-0224-T. Epub 2014 Jan 14. Mol Cancer Res. 2014. PMID: 24425783 Free PMC article.
-
KDM5B Promotes Drug Resistance by Regulating Melanoma-Propagating Cell Subpopulations.Mol Cancer Ther. 2019 Mar;18(3):706-717. doi: 10.1158/1535-7163.MCT-18-0395. Epub 2018 Dec 6. Mol Cancer Ther. 2019. PMID: 30523048 Free PMC article.
-
Targeting CD20 in melanoma patients at high risk of disease recurrence.Mol Ther. 2012 May;20(5):1056-62. doi: 10.1038/mt.2012.27. Epub 2012 Feb 21. Mol Ther. 2012. PMID: 22354376 Free PMC article. Clinical Trial.
-
Regulation of viability, differentiation and death of human melanoma cells carrying neural stem cell biomarkers: a possibility for neural trans-differentiation.Apoptosis. 2015 Jul;20(7):996-1015. doi: 10.1007/s10495-015-1131-3. Apoptosis. 2015. PMID: 25953317 Free PMC article.
-
Effect of melanoma stem cells on melanoma metastasis.Oncol Lett. 2021 Jul;22(1):566. doi: 10.3892/ol.2021.12827. Epub 2021 May 29. Oncol Lett. 2021. PMID: 34113394 Free PMC article. Review.
References
-
- Clarke MF, Dicks JE, Dirks PB, et al. Cancer stem cells—perspectives on current status and future directions: AACR Workshop on cancer stem cells. Cancer Res. 2006;66:9339–44. - PubMed
-
- Visvader JE, Lindeman GJ. Cancer stem cells in solid tumors: accumulating evidence and unresolved questions. Nat Rev Cancer. 2008;8:755–68. - PubMed
-
- Lapidot T, Sirard C, Vormoor J, et al. A cell initiating human acute myeloid leukaemia after transplantation into SCID mice. Nature. 1994;367:645–8. - PubMed
-
- Singh SK, Clarke ID, Terasaki M, et al. Identification of a cancer stem cell in human brain tumors. Cancer Res. 2003;63:5821–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

