Expression of the alternative oxidase complements cytochrome c oxidase deficiency in human cells
- PMID: 20049701
- PMCID: PMC3378104
- DOI: 10.1002/emmm.200900001
Expression of the alternative oxidase complements cytochrome c oxidase deficiency in human cells
Abstract
Cytochrome c oxidase (COX) deficiency is associated with a wide spectrum of clinical conditions, ranging from early onset devastating encephalomyopathy and cardiomyopathy, to neurological diseases in adulthood and in the elderly. No method of compensating successfully for COX deficiency has been reported so far. In vitro, COX-deficient human cells require additional glucose, pyruvate and uridine for normal growth and are specifically sensitive to oxidative stress. Here, we have tested whether the expression of a mitochondrially targeted, cyanide-resistant, alternative oxidase (AOX) from Ciona intestinalis could alleviate the metabolic abnormalities of COX-deficient human cells either from a patient harbouring a COX15 pathological mutation or rendered deficient by silencing the COX10 gene using shRNA. We demonstrate that the expression of the AOX, well-tolerated by the cells, compensates for both the growth defect and the pronounced oxidant-sensitivity of COX-deficient human cells.
Figures
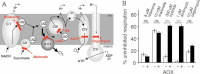
The cyt pathway bypass provided by AOX and the targets of different respiratory chain inhibitors. Aa, antimycin; c, cyt c; CI–V, the various respiratory chain complexes; im, inner membrane; UQ, ubiquinone.
The effect of AOX expression on respiration of HEK-293 cells treated with various OXPHOS inhibitors.

COX activity in control (AOX-transgenic) and HEK-293 cells expressing COX10-targeted shRNA (clone selected after transfection with pSM2C-COX10 construct). Values are means of triplicates performed on three different clones. Inset shows immunoblot of 100 µg total cell lysates. Specificity of the anti-COX10 shRNA was established by using a scrambled shRNA which did not affect COX10 level or COX activity (data not shown).
Cell growth of either control (AOX-transgenic) or COX10− HEK-293 cells, with or without doxycyclin induction of AOX expression, in non-selective medium. Cells were seeded at 10,000 cells/cm2.
Number of cells after 7 days of growth (end of the exponential growth phase) of either control (AOX-transgenic) or COX10− cells, with or without doxycycline induction of AOX expression, in selective medium (i.e. 1 g/l glucose).
Respiration of intact HEK control and COX10− cells ± AOX. KCN was 300 µM, n-propyl gallate (n-PG) 50 µM. Numbers along the traces represent nmol O2/min per mg protein (mean values from three experiments).
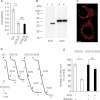
COX and SCCR activity ratios in immortalized control, COX15− and COX15−: AOX fibroblasts.
Respiration of intact control, COX15− and COX15−: AOX cells measured using a Clark electrode. KCN was 300 µM, n-PG 50µM. Numbers along the traces represent nmol O2/min per mg protein (mean values from three experiments).
Immunoblot of 25 µg whole cell lysates from COX15-mutant fibroblasts transduced (or not) with AOX, probed with antibodies against lily AOX (lanes 1 and 2), and actin (lanes 3 and 4).
AOX mitochondrial localization assessed by AOX antibody. Scale bar, 20 µm.
Numbers of cells after 7 day growth in selective or non-selective media, expressed as percentage of cell number obtained under non-selective condition. Cells seeded at 15,000 cells/cm2.
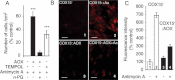
Growth of AOX-expressing or non-expressing cells (3 d) in low glucose medium in the presence or absence of 5 µM antimycin ±1 mM TEMPOL (4-hydroxy-2,2,6,6-tetramethylpiperidinyloxy) (percentage of cells in the absence of antimycin). Cells seeded at 15,000 cells/cm2.
Fluorescence microscopy images using MitoSOX™ probe, with or without 5 µM antimycin (30 min). Panels 1 and 2, COX15− cells (1 untreated; 2 antimycin treated), Panels 3 and 4, COX15− : AOX cells (3 untreated; 4 antimycin treated) (scale bars represent 40 µm).
Quantification of MitoSOX™ staining (Fig 3B panels 1–4) expressed as a percentage of the labelling measured in control COX15− cells (panel 1).
Similar articles
-
The alternative oxidase, a tool for compensating cytochrome c oxidase deficiency in human cells.Physiol Plant. 2009 Dec;137(4):427-34. doi: 10.1111/j.1399-3054.2009.01248.x. Epub 2009 Apr 25. Physiol Plant. 2009. PMID: 19493305
-
Expression of the Ciona intestinalis alternative oxidase (AOX) in Drosophila complements defects in mitochondrial oxidative phosphorylation.Cell Metab. 2009 May;9(5):449-60. doi: 10.1016/j.cmet.2009.03.004. Cell Metab. 2009. PMID: 19416715
-
Expression of the alternative oxidase mitigates beta-amyloid production and toxicity in model systems.Free Radic Biol Med. 2016 Jul;96:57-66. doi: 10.1016/j.freeradbiomed.2016.04.006. Epub 2016 Apr 14. Free Radic Biol Med. 2016. PMID: 27094492
-
Human cytochrome oxidase deficiency.Pediatr Res. 2000 Nov;48(5):581-5. doi: 10.1203/00006450-200011000-00004. Pediatr Res. 2000. PMID: 11044474 Review.
-
Tissue-specific adaptations to cytochrome c oxidase deficiency shape physiological outcomes.Biochim Biophys Acta Mol Basis Dis. 2025 Mar;1871(3):167567. doi: 10.1016/j.bbadis.2024.167567. Epub 2024 Nov 28. Biochim Biophys Acta Mol Basis Dis. 2025. PMID: 39613003 Review.
Cited by
-
Alternative oxidase encoded by sequence-optimized and chemically-modified RNA transfected into mammalian cells is catalytically active.Gene Ther. 2022 Dec;29(12):655-664. doi: 10.1038/s41434-021-00235-z. Epub 2021 Mar 4. Gene Ther. 2022. PMID: 33664504 Free PMC article.
-
Mitochondrial medicine: to a new era of gene therapy for mitochondrial DNA mutations.J Inherit Metab Dis. 2011 Apr;34(2):327-44. doi: 10.1007/s10545-010-9131-5. Epub 2010 Jun 23. J Inherit Metab Dis. 2011. PMID: 20571866 Review.
-
Development of pharmacological strategies for mitochondrial disorders.Br J Pharmacol. 2014 Apr;171(8):1798-817. doi: 10.1111/bph.12456. Br J Pharmacol. 2014. PMID: 24116962 Free PMC article. Review.
-
Alternative oxidase expression in the mouse enables bypassing cytochrome c oxidase blockade and limits mitochondrial ROS overproduction.PLoS Genet. 2013;9(1):e1003182. doi: 10.1371/journal.pgen.1003182. Epub 2013 Jan 3. PLoS Genet. 2013. PMID: 23300486 Free PMC article.
-
Developmental arrest in Drosophila melanogaster caused by mitochondrial DNA replication defects cannot be rescued by the alternative oxidase.Sci Rep. 2018 Jul 18;8(1):10882. doi: 10.1038/s41598-018-29150-x. Sci Rep. 2018. PMID: 30022066 Free PMC article.
References
-
- Barrientos A, Barros MH, Valnot I, Rotig A, Rustin P, Tzagoloff A. Cytochrome oxidase in health and disease. Gene. 2002;286:53–63. - PubMed
-
- Benit P, Goncalves S, Philippe Dassa E, Briere JJ, Martin G, Rustin P. Three spectrophotometric assays for the measurement of the five respiratory chain complexes in minuscule biological samples. Clin Chim Acta. 2006;374:81–88. - PubMed
-
- Bovia F, Salmon P, Matthes T, Kvell K, Nguyen TH, Werner-Favre C, Barnet M, Nagy M, Leuba F, Arrighi JF, Piguet V, Trono D, Zubler RH. Efficient transduction of primary human B lymphocytes and nondividing myeloma B cells with HIV-1-derived lentiviral vectors. Blood. 2003;101:1727–1733. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

