The function of CCR3 on mouse bone marrow-derived mast cells in vitro
- PMID: 20050333
- PMCID: PMC2807492
- DOI: 10.1111/j.1365-2567.2009.03151.x
The function of CCR3 on mouse bone marrow-derived mast cells in vitro
Abstract
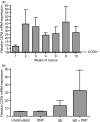
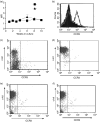
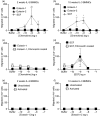
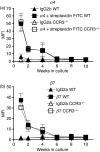
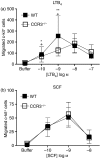
The mechanisms governing the population of tissues by mast cells are not fully understood, but several studies using human mast cells have suggested that expression of the chemokine receptor CCR3 and migration to its ligands may be important. In CCR3-deficient mice, a change in mast cell tissue distribution in the airways following allergen challenge was reported compared with wild-type mice. In addition, there is evidence that CCR3 is important in mast cell maturation in mouse. In this study, bone marrow-derived mast cells (BMMCs) were cultured and CCR3 expression and the migratory response to CCR3 ligands were characterized. In addition, BMMCs were cultured from wild-type and CCR3-deficient mice and their phenotype and migratory responses were compared. CCR3 messenger RNA was detectable in BMMCs, but this was not significantly increased after activation by immunoglobulin E (IgE). CCR3 protein was not detected on BMMCs during maturation and expression could not be enhanced after IgE activation. Resting and IgE-activated immature and mature BMMCs did not migrate in response to the CCR3 ligands eotaxin- 1 and eotaxin-2. Comparing wild-type and CCR3-deficient BMMCs, there were no differences in mast cell phenotype or ability to migrate to the mast cell chemoattractants leukotriene B4 and stem cell factor. The results of this study show that CCR3 may not mediate mast cell migration in mouse BMMCs in vitro. These observations need to be considered in relation to the findings of CCR3 deficiency on mast cells in vivo.
Figures

References
-
- Sayed BA, Christy A, Quirion MR, Brown MA. The master switch: the role of mast cells in autoimmunity and tolerance. Annu Rev Immunol. 2008;26:705–39. - PubMed
-
- Fokkens WJ, Godthelp T, Holm AF, Blom H, Mulder PG, Vroom TM, Rijntjes E. Dynamics of mast cells in the nasal mucosa of patients with allergic rhinitis and non-allergic controls: a biopsy study. Clin Exp Allergy. 1992;22:701–10. - PubMed
-
- Nouri-Aria KT, Pilette C, Jacobson MR, Watanabe H, Durham SR. IL-9 and c-Kit+ mast cells in allergic rhinitis during seasonal allergen exposure: effect of immunotherapy. J Allergy Clin Immunol. 2005;116:73–9. - PubMed
-
- Brightling CE, Bradding P, Symon FA, Holgate ST, Wardlaw AJ, Pavord ID. Mast-cell infiltration of airway smooth muscle in asthma. N Engl J Med. 2002;346:1699–705. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

