KChIP2 attenuates cardiac hypertrophy through regulation of Ito and intracellular calcium signaling
- PMID: 20051248
- PMCID: PMC2866822
- DOI: 10.1016/j.yjmcc.2009.12.019
KChIP2 attenuates cardiac hypertrophy through regulation of Ito and intracellular calcium signaling
Abstract
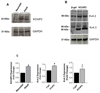
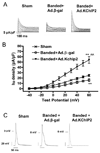
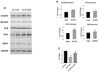
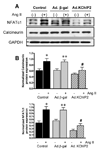
Recent evidence shows that the auxiliary subunit KChIP2, which assembles with pore-forming Kv4-subunits, represents a new potential regulator of the cardiac calcium-independent transient outward potassium current (I(to)) density. In hypertrophy and heart failure, KChIP2 expression has been found to be significantly decreased. Our aim was to examine the role of KChIP2 in cardiac hypertrophy and the effect of restoring its expression on electrical remodeling and cardiac mechanical function using a combination of molecular, biochemical and gene targeting approaches. KChIP2 overexpression through gene transfer of Ad.KChIP2 in neonatal cardiomyocytes resulted in a significant increase in I(to)-channel forming Kv4.2 and Kv4.3 protein levels. In vivo gene transfer of KChIP2 in aortic banded adult rats showed that, compared to sham-operated or Ad.beta-gal-transduced hearts, KChIP2 significantly attenuated the developed left ventricular hypertrophy, robustly increased I(to) densities, shortened action potential duration, and significantly altered myocyte mechanics by shortening contraction amplitudes and maximal rates of contraction and relaxation velocities and decreasing Ca(2+) transients. Interestingly, blocking I(to) with 4-aminopyridine in KChIP2-overexpressing adult cardiomyocytes significantly increased the Ca(2+) transients to control levels. One-day-old rat pups intracardially transduced with KChIP2 for two months then subjected to aortic banding for 6-8 weeks (to induce hypertrophy) showed similar echocardiographic, electrical and mechanical remodeling parameters. In addition, in cultured adult cardiomyocytes, KChIP2 overexpression increased the expression of Ca(2+)-ATPase (SERCA2a) and sodium calcium exchanger but had no effect on ryanodine receptor 2 or phospholamban expression. In neonatal myocytes, KChIP2 notably reversed Ang II-induced hypertrophic changes in protein synthesis and MAP-kinase activation. It also significantly decreased calcineurin expression, NFATc1 expression and nuclear translocation and its downstream target, MCiP1.4. Altogether, these data show that KChIP2 can attenuate cardiac hypertrophy possibly through modulation of intracellular calcium concentration and calcineurin/NFAT pathway.
(c) 2009. Published by Elsevier Ltd. All rights reserved.
Figures







Similar articles
-
MG53, A Novel Regulator of KChIP2 and Ito,f, Plays a Critical Role in Electrophysiological Remodeling in Cardiac Hypertrophy.Circulation. 2019 Apr 30;139(18):2142-2156. doi: 10.1161/CIRCULATIONAHA.118.029413. Circulation. 2019. PMID: 30760025
-
KChIP2 regulates the cardiac Ca2+ transient and myocyte contractility by targeting ryanodine receptor activity.PLoS One. 2017 Apr 6;12(4):e0175221. doi: 10.1371/journal.pone.0175221. eCollection 2017. PLoS One. 2017. PMID: 28384221 Free PMC article.
-
Prolonged leptin treatment increases transient outward K⁺ current via upregulation of Kv4.2 and Kv4.3 channel subunits in adult rat ventricular myocytes.Pflugers Arch. 2014 May;466(5):903-14. doi: 10.1007/s00424-013-1348-3. Epub 2013 Sep 18. Pflugers Arch. 2014. PMID: 24046152
-
TRP Channels in the Heart.In: Emir TLR, editor. Neurobiology of TRP Channels. Boca Raton (FL): CRC Press/Taylor & Francis; 2017. Chapter 9. In: Emir TLR, editor. Neurobiology of TRP Channels. Boca Raton (FL): CRC Press/Taylor & Francis; 2017. Chapter 9. PMID: 29356479 Free Books & Documents. Review.
-
Nuclear calcium in cardiac myocytes.J Cardiovasc Pharmacol. 2015 Mar;65(3):211-7. doi: 10.1097/FJC.0000000000000174. J Cardiovasc Pharmacol. 2015. PMID: 25329749 Free PMC article. Review.
Cited by
-
Genetic architecture of microRNA expression: implications for the transcriptome and complex traits.Am J Hum Genet. 2012 Jun 8;90(6):1046-63. doi: 10.1016/j.ajhg.2012.04.023. Epub 2012 May 31. Am J Hum Genet. 2012. PMID: 22658545 Free PMC article.
-
A personalized, multiomics approach identifies genes involved in cardiac hypertrophy and heart failure.NPJ Syst Biol Appl. 2018 Feb 24;4:12. doi: 10.1038/s41540-018-0046-3. eCollection 2018. NPJ Syst Biol Appl. 2018. PMID: 29507758 Free PMC article.
-
Calcium/calmodulin-dependent protein kinase II associates with the K+ channel isoform Kv4.3 in adult rat optic nerve.Front Neuroanat. 2022 Sep 8;16:958986. doi: 10.3389/fnana.2022.958986. eCollection 2022. Front Neuroanat. 2022. PMID: 36172564 Free PMC article.
-
Impact of KChIP2 on Cardiac Electrophysiology and the Progression of Heart Failure.Front Physiol. 2012 May 4;3:118. doi: 10.3389/fphys.2012.00118. eCollection 2012. Front Physiol. 2012. PMID: 22586403 Free PMC article.
-
The Ca2+ transient as a feedback sensor controlling cardiomyocyte ionic conductances in mouse populations.Elife. 2018 Sep 25;7:e36717. doi: 10.7554/eLife.36717. Elife. 2018. PMID: 30251624 Free PMC article.
References
-
- Nabauer M, Kaab S. Potassium channel down-regulation in heart failure. Cardiovascular research. 1998 Feb;37(2):324–334. - PubMed
-
- Tomaselli GF, Marban E. Electrophysiological remodeling in hypertrophy and heart failure. Cardiovascular research. 1999 May;42(2):270–283. - PubMed
-
- Wickenden AD, Kaprielian R, Kassiri Z, Tsoporis JN, Tsushima R, Fishman GI, et al. The role of action potential prolongation and altered intracellular calcium handling in the pathogenesis of heart failure. Cardiovascular research. 1998 Feb;37(2):312–323. - PubMed
-
- Gidh-Jain M, Huang B, Jain P, el-Sherif N. Differential expression of voltage-gated K+ channel genes in left ventricular remodeled myocardium after experimental myocardial infarction. Circulation research. 1996 Oct;79(4):669–675. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

