Persistent induction of cytochrome P4501A1 in human hepatoma cells by 3-methylcholanthrene: evidence for sustained transcriptional activation of the CYP1A1 promoter
- PMID: 20051482
- PMCID: PMC2846024
- DOI: 10.1124/jpet.109.162222
Persistent induction of cytochrome P4501A1 in human hepatoma cells by 3-methylcholanthrene: evidence for sustained transcriptional activation of the CYP1A1 promoter
Abstract
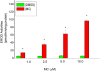
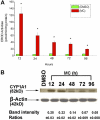
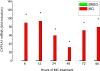
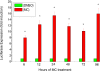
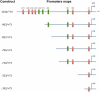
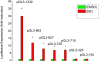
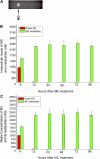
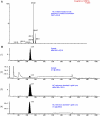
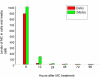
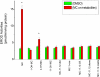
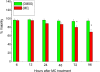
Cytochrome P450 (P450)1A1 plays a critical role in the metabolic activation and detoxification of polycyclic aromatic hydrocarbons (PAHs), many of which are potent human carcinogens. In this investigation, we tested the hypothesis that MC elicits persistent induction of CYP1A1 expression in human hepatoma cells (HepG2) and that this phenomenon is mediated by sustained transcriptional activation of the CYP1A1 promoter. Treatment of HepG2 cells with MC resulted in marked induction (8-20-fold) of ethoxyresorufin O-de-ethylase activities, CYP1A1 apoprotein contents, and mRNA levels, which persisted for up to 96 h. MC also caused sustained transcriptional activation of the human CYP1A1 promoter for up to 96 h, as inferred from transient transfection experiments. Experiments with deletion constructs indicated that Ah response elements located at -886, -974, and -1047, but not -491, nucleotides from the start site, contributed to the sustained transcriptional activation of the CYP1A1 promoter. Electrophoretic mobility-shift and chromatin immunoprecipitation assays suggested that prolonged CYP1A1 induction was mediated by Ah receptor (AHR)-independent mechanisms. Experiments with [3H]MC and liquid chromatography-tandem mass spectrometry demonstrated rapid elimination of MC and its metabolites from the cells by 12 to 24 h, suggesting that these compounds did not elicit sustained CYP1A1 induction via the classical AHR-mediated pathway. In conclusion, the results of this study support the hypothesis that MC causes persistent induction of CYP1A1 in human hepatoma cells by mechanisms entailing sustained transcriptional activation of the CYP1A1 promoter via AHR-independent mechanisms. These observations have important implications for human carcinogenesis mediated by PAHs.
Figures

References
-
- Boobis AR, Nebert DW, Felton JS. (1977) Comparison of beta-naphthoflavone and 3-methylcholanthrene as inducers of hepatic cytochrome(s) P-448 and aryl hydrocarbon (benzo[a]pyrene) hydroxylase activity. Mol Pharmacol 13:259–268 - PubMed
-
- Conney AH. (1986) Induction of microsomal cytochrome P-450 enzymes: the first Bernard B. Brodie lecture at Pennsylvania State University. Life Sci 39:2493–2518 - PubMed
-
- Coumoul X, Diry M, Robillot C, Barouki R. (2001) Differential regulation of cytochrome P450 1A1 and 1B1 by a combination of dioxin and pesticides in the breast tumor cell line MCF-7. Cancer Res 61:3942–3948 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

