Zebrafish (Danio rerio) gill neuroepithelial cells are sensitive chemoreceptors for environmental CO2
- PMID: 20051495
- PMCID: PMC2834944
- DOI: 10.1113/jphysiol.2009.184739
Zebrafish (Danio rerio) gill neuroepithelial cells are sensitive chemoreceptors for environmental CO2
Abstract
Adult zebrafish exhibit hyperventilatory responses to absolute environmental CO(2) levels as low as 0.13% ( mmHg), more than an order of magnitude lower than the typical arterial levels (40 mmHg) monitored by the mammalian carotid body. The sensory basis underlying the ability of fish to detect and respond to low ambient CO(2) levels is not clear. Here, we show that the neuroepithelial cells (NECs) of the zebrafish gill, known to sense O(2) levels, also respond to low levels of CO(2). An electrophysiological characterization of this response using both current and voltage clamp protocols revealed that for increasing CO(2) levels, a background K(+) channel was inhibited, resulting in a partial pressure-dependent depolarization of the NEC. To elucidate the signalling pathway underlying K(+) channel inhibition, we used immunocytochemistry to show that these NECs express carbonic anhydrase (CA), an enzyme involved in CO(2) sensing in the mammalian carotid body. Further, the NEC response to CO(2) (magnitude of membrane depolarization and time required to achieve maximal response), under conditions of constant pH, was reduced by 50% by the CA-inhibitor acetazolamide. This suggests that the CO(2) detection mechanism involves an intracellular sensor that is responsive to the rate of acidification associated with the hydration of CO(2) and which does not require a change of extracellular pH. Because some cells that were responsive to increasing also responded to hypoxia with membrane depolarization, the present results demonstrate that a subset of the NECs in the zebrafish gill are bimodal sensors of CO(2) and O(2).
Figures
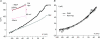
 reverses near EK (calculated using bath and pipette K+ concentrations) and fits the corresponding Goldman–Hodgkin–Katz current equation (GHK, continuous curve; Hille, 2001), suggesting that it is predominantly carried by K+ ions.
reverses near EK (calculated using bath and pipette K+ concentrations) and fits the corresponding Goldman–Hodgkin–Katz current equation (GHK, continuous curve; Hille, 2001), suggesting that it is predominantly carried by K+ ions.
 mmHg, dashed line), respectively. C, current clamp recording showing depolarization during perfusion with hypercapnic (filled arrow bar) or hypoxic solution (dashed bar) measured on the same cell. D, mean ±
mmHg, dashed line), respectively. C, current clamp recording showing depolarization during perfusion with hypercapnic (filled arrow bar) or hypoxic solution (dashed bar) measured on the same cell. D, mean ±
 mmHg), 0.5% (
mmHg), 0.5% ( mmHg) or 1% (
mmHg) or 1% ( mmHg) CO2. Scale bar = 10 mV/10 s in C (applies to A and B as well). D, mean ±
mmHg) CO2. Scale bar = 10 mV/10 s in C (applies to A and B as well). D, mean ±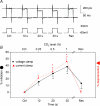

Similar articles
-
CO(2) signaling in chemosensory neuroepithelial cells of the zebrafish gill filaments: role of intracellular Ca(2+) and pH.Adv Exp Med Biol. 2012;758:143-8. doi: 10.1007/978-94-007-4584-1_20. Adv Exp Med Biol. 2012. PMID: 23080155
-
Neuroepithelial oxygen chemoreceptors of the zebrafish gill.J Physiol. 2004 Nov 1;560(Pt 3):737-52. doi: 10.1113/jphysiol.2004.069294. Epub 2004 Aug 26. J Physiol. 2004. PMID: 15331683 Free PMC article.
-
Mechanisms and consequences of carbon dioxide sensing in fish.Respir Physiol Neurobiol. 2012 Dec 1;184(3):309-15. doi: 10.1016/j.resp.2012.06.013. Epub 2012 Jun 15. Respir Physiol Neurobiol. 2012. PMID: 22705499 Review.
-
Extracellular H+ induces Ca2+ signals in respiratory chemoreceptors of zebrafish.Pflugers Arch. 2015 Feb;467(2):399-413. doi: 10.1007/s00424-014-1514-2. Epub 2014 Apr 26. Pflugers Arch. 2015. PMID: 24770973
-
Peripheral chemoreceptors in fish: A brief history and a look ahead.Comp Biochem Physiol A Mol Integr Physiol. 2015 Aug;186:27-38. doi: 10.1016/j.cbpa.2014.09.002. Epub 2014 Sep 16. Comp Biochem Physiol A Mol Integr Physiol. 2015. PMID: 25218943 Review.
Cited by
-
A marine teleost, Opsanus beta, compensates acidosis in hypersaline water by H+ excretion or reduced HCO3- excretion rather than HCO3- uptake.J Comp Physiol B. 2021 Jan;191(1):85-98. doi: 10.1007/s00360-020-01320-2. Epub 2020 Oct 17. J Comp Physiol B. 2021. PMID: 33070210
-
Carbon dioxide-sensing in organisms and its implications for human disease.Cell Mol Life Sci. 2014 Mar;71(5):831-45. doi: 10.1007/s00018-013-1470-6. Epub 2013 Sep 18. Cell Mol Life Sci. 2014. PMID: 24045706 Free PMC article. Review.
-
Effects of Water Acidification on Senegalese Sole Solea senegalensis Health Status and Metabolic Rate: Implications for Immune Responses and Energy Use.Front Physiol. 2020 Jan 31;11:26. doi: 10.3389/fphys.2020.00026. eCollection 2020. Front Physiol. 2020. PMID: 32082190 Free PMC article.
-
Zebrafish (Danio rerio) behaviour is largely unaffected by elevated pCO2.Conserv Physiol. 2016 Dec 29;4(1):cow065. doi: 10.1093/conphys/cow065. eCollection 2016. Conserv Physiol. 2016. PMID: 28066551 Free PMC article.
-
Peripheral chemoreceptors: function and plasticity of the carotid body.Compr Physiol. 2012 Jan;2(1):141-219. doi: 10.1002/cphy.c100069. Compr Physiol. 2012. PMID: 23728973 Free PMC article. Review.
References
-
- Bailly Y, Dunelerb S, Laurent P. The neuroepithelial cells of the fish gill filament: indolamine-immunocyto-chemistry and innervation. Anat Rec. 1992;233:143–161. - PubMed
-
- Bayliss DA, Sirois JE, Talley EM. The TASK family: two-pore domain background K+ channels. Mol Interv. 2003;3:205–219. - PubMed
-
- Bayliss DA, Talley EM, Sirois JE, Lei Q. TASK-1 is a highly modulated pH-sensitive ‘leak’ K+ channel expressed in brainstem respiratory neurons. Respir Physiol. 2001;129:159–174. - PubMed
-
- Buckler KJ. TASK-like potassium channels and oxygen sensing in the carotid body. Respir Physiol Neurobiol. 2007;157:55–64. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

