Ascorbic acid prevents blood-brain barrier disruption and sensory deficit caused by sustained compression of primary somatosensory cortex
- PMID: 20051973
- PMCID: PMC2949198
- DOI: 10.1038/jcbfm.2009.277
Ascorbic acid prevents blood-brain barrier disruption and sensory deficit caused by sustained compression of primary somatosensory cortex
Abstract
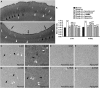
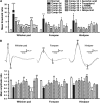
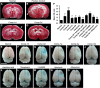
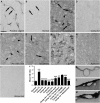
Transient compression of rat somatosensory cortex has been reported to affect cerebral microvasculature and sensory function simultaneously. However, the effects of long-term cortical compression remain unknown. Here, we investigated whether and to what extent sustained but moderate epidural compression of rat somatosensory cortex impairs somatic sensation and/or cortical microvasculature. Electrophysiological and behavioral tests revealed that sustained compression caused only short-term sensory deficit, particularly at 1 day after injury. Although the diameter of cortical microvessels was coincidentally reduced, no ischemic insult was observed. By measuring Evans Blue and immunoglobulin G extravasation, the blood-brain barrier (BBB) permeability was found to dramatically increase during 1 to 3 days, but this did not lead to brain edema. Furthermore, immunoblotting showed that the BBB component proteins occludin, claudin-5, type IV collagen, and glial fibrillary acidic protein were markedly upregulated in the injured cortex during 1 to 2 weeks when BBB regained integrity. Conversely, treatment of ascorbic acid prevented compression-induced BBB disruption and sensory impairment. Together, these data suggest that sustained compression of the somatosensory cortex compromises BBB integrity and somatic sensation only in the early period. Ascorbic acid may be used therapeutically to modulate cortical compression and/or BBB dysfunction.
Figures


Similar articles
-
Removal of a compressive mass causes a transient disruption of blood-brain barrier but a long-term recovery of spiny stellate neurons in the rat somatosensory cortex.Restor Neurol Neurosci. 2021;39(2):111-127. doi: 10.3233/RNN-201085. Restor Neurol Neurosci. 2021. PMID: 34024792
-
Erythropoietin attenuates intracerebral hemorrhage by diminishing matrix metalloproteinases and maintaining blood-brain barrier integrity in mice.Acta Neurochir Suppl. 2008;105:105-12. doi: 10.1007/978-3-211-09469-3_22. Acta Neurochir Suppl. 2008. PMID: 19066093
-
Lanthanum Chloride Impairs the Blood-Brain Barrier Integrity by Reduction of Junctional Proteins and Upregulation of MMP-9 in Rats.Biol Trace Elem Res. 2019 Feb;187(2):482-491. doi: 10.1007/s12011-018-1402-2. Epub 2018 Jun 6. Biol Trace Elem Res. 2019. PMID: 29876795
-
Simvastatin attenuates sepsis-induced blood-brain barrier integrity loss.J Surg Res. 2015 Apr;194(2):591-598. doi: 10.1016/j.jss.2014.11.030. Epub 2014 Nov 26. J Surg Res. 2015. PMID: 25534234
-
Increased caveolin-1 expression precedes decreased expression of occludin and claudin-5 during blood-brain barrier breakdown.Acta Neuropathol. 2007 Nov;114(5):459-69. doi: 10.1007/s00401-007-0274-x. Epub 2007 Aug 9. Acta Neuropathol. 2007. PMID: 17687559
Cited by
-
Reliability of VEP Recordings Using Chronically Implanted Screw Electrodes in Mice.Transl Vis Sci Technol. 2015 Apr 28;4(2):15. doi: 10.1167/tvst.4.2.15. eCollection 2015 Apr. Transl Vis Sci Technol. 2015. PMID: 25938003 Free PMC article.
-
Neuro-Inflammation Modulation and Post-Traumatic Brain Injury Lesions: From Bench to Bed-Side.Int J Mol Sci. 2022 Sep 23;23(19):11193. doi: 10.3390/ijms231911193. Int J Mol Sci. 2022. PMID: 36232495 Free PMC article. Review.
-
Cortical spheroids show strain-dependent cell viability loss and neurite disruption following sustained compression injury.PLoS One. 2024 Aug 19;19(8):e0295086. doi: 10.1371/journal.pone.0295086. eCollection 2024. PLoS One. 2024. PMID: 39159236 Free PMC article.
-
Chronic cerebrovascular dysfunction after traumatic brain injury.J Neurosci Res. 2016 Jul;94(7):609-22. doi: 10.1002/jnr.23732. Epub 2016 Apr 27. J Neurosci Res. 2016. PMID: 27117494 Free PMC article. Review.
-
Poly(ε-caprolactone)-block-poly(ethyl ethylene phosphate) micelles for brain-targeting drug delivery: in vitro and in vivo valuation.Pharm Res. 2010 Dec;27(12):2657-69. doi: 10.1007/s11095-010-0265-9. Epub 2010 Sep 18. Pharm Res. 2010. PMID: 20848303
References
-
- Abbott NJ, Ronnback L, Hansson E. Astrocyte-endothelial interactions at the blood-brain barrier. Nat Rev Neurosci. 2006;7:41–53. - PubMed
-
- Andras IE, Deli MA, Veszelka S, Hayashi K, Hennig B, Toborek M. The NMDA and AMPA/KA receptors are involved in glutamate-induced alterations of occludin expression and phosphorylation in brain endothelial cells. J Cereb Blood Flow Metab. 2007;27:1431–1443. - PubMed
-
- Boje KM, Lakhman SS. Nitric oxide redox species exert differential permeability effects on the blood-brain barrier. J Pharmacol Exp Ther. 2000;293:545–550. - PubMed
-
- Burnett MG, Detre JA, Greenberg JH. Activation-flow coupling during graded cerebral ischemia. Brain Res. 2005;1047:112–118. - PubMed
-
- Chen JR, Wang YJ, Tseng GF. The effect of epidural compression on cerebral cortex: a rat model. J Neurotrauma. 2003;20:767–780. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

