Presynaptic external calcium signaling involves the calcium-sensing receptor in neocortical nerve terminals
- PMID: 20052292
- PMCID: PMC2797309
- DOI: 10.1371/journal.pone.0008563
Presynaptic external calcium signaling involves the calcium-sensing receptor in neocortical nerve terminals
Abstract
Background: Nerve terminal invasion by an axonal spike activates voltage-gated channels, triggering calcium entry, vesicle fusion, and release of neurotransmitter. Ion channels activated at the terminal shape the presynaptic spike and so regulate the magnitude and duration of calcium entry. Consequently characterization of the functional properties of ion channels at nerve terminals is crucial to understand the regulation of transmitter release. Direct recordings from small neocortical nerve terminals have revealed that external [Ca(2+)] ([Ca(2+)](o)) indirectly regulates a non-selective cation channel (NSCC) in neocortical nerve terminals via an unknown [Ca(2+)](o) sensor. Here, we identify the first component in a presynaptic calcium signaling pathway.
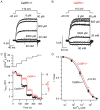
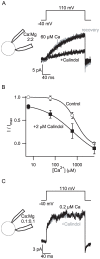
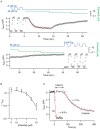
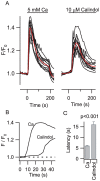
Methodology/principal findings: By combining genetic and pharmacological approaches with direct patch-clamp recordings from small acutely isolated neocortical nerve terminals we identify the extracellular calcium sensor. Our results show that the calcium-sensing receptor (CaSR), a previously identified G-protein coupled receptor that is the mainstay in serum calcium homeostasis, is the extracellular calcium sensor in these acutely dissociated nerve terminals. The NSCC currents from reduced function mutant CaSR mice were less sensitive to changes in [Ca(2+)](o) than wild-type. Calindol, an allosteric CaSR agonist, reduced NSCC currents in direct terminal recordings in a dose-dependent and reversible manner. In contrast, glutamate and GABA did not affect the NSCC currents.
Conclusions/significance: Our experiments identify CaSR as the first component in the [Ca(2+)](o) sensor-NSCC signaling pathway in neocortical terminals. Decreases in [Ca(2+)](o) will depress synaptic transmission because of the exquisite sensitivity of transmitter release to [Ca(2+)](o) following its entry via voltage-activated Ca(2+) channels. CaSR may detects such falls in [Ca(2+)](o) and increase action potential duration by increasing NSCC activity, thereby attenuating the impact of decreases in [Ca(2+)](o) on release probability. CaSR is positioned to detect the dynamic changes of [Ca(2+)](o) and provide presynaptic feedback that will alter brain excitability.
Conflict of interest statement
Figures

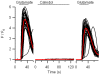
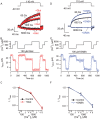
Similar articles
-
Calcium-sensing receptor activation depresses synaptic transmission.J Neurosci. 2008 Nov 12;28(46):12062-70. doi: 10.1523/JNEUROSCI.4134-08.2008. J Neurosci. 2008. PMID: 19005071 Free PMC article.
-
Recordings from single neocortical nerve terminals reveal a nonselective cation channel activated by decreases in extracellular calcium.Neuron. 2004 Jan 22;41(2):243-56. doi: 10.1016/s0896-6273(03)00837-7. Neuron. 2004. PMID: 14741105
-
Calcium-sensing receptor: a high-affinity presynaptic target for aminoglycoside-induced weakness.Neuropharmacology. 2009 Oct-Nov;57(5-6):502-5. doi: 10.1016/j.neuropharm.2009.07.031. Epub 2009 Jul 29. Neuropharmacology. 2009. PMID: 19646457 Free PMC article.
-
Ca2+-sensing receptor-TRP channel-mediated Ca2+ signaling: Functional diversity and pharmacological complexity.Eur J Pharmacol. 2024 Aug 15;977:176717. doi: 10.1016/j.ejphar.2024.176717. Epub 2024 Jun 8. Eur J Pharmacol. 2024. PMID: 38857682 Review.
-
International Union of Basic and Clinical Pharmacology. CVIII. Calcium-Sensing Receptor Nomenclature, Pharmacology, and Function.Pharmacol Rev. 2020 Jul;72(3):558-604. doi: 10.1124/pr.119.018531. Pharmacol Rev. 2020. PMID: 32467152 Free PMC article. Review.
Cited by
-
Apolipoprotein E4 Effects a Distinct Transcriptomic Profile and Dendritic Arbor Characteristics in Hippocampal Neurons Cultured in vitro.Front Aging Neurosci. 2022 Apr 29;14:845291. doi: 10.3389/fnagi.2022.845291. eCollection 2022. Front Aging Neurosci. 2022. PMID: 35572125 Free PMC article.
-
High calcium concentrations reduce cellular excitability of mouse MNTB neurons.Brain Res. 2023 Dec 1;1820:148568. doi: 10.1016/j.brainres.2023.148568. Epub 2023 Sep 7. Brain Res. 2023. PMID: 37689332 Free PMC article.
-
Calcium regulation of spontaneous and asynchronous neurotransmitter release.Cell Calcium. 2012 Sep-Oct;52(3-4):226-33. doi: 10.1016/j.ceca.2012.06.001. Epub 2012 Jun 29. Cell Calcium. 2012. PMID: 22748761 Free PMC article.
-
Spontaneous glutamate release is independent of calcium influx and tonically activated by the calcium-sensing receptor.J Neurosci. 2011 Mar 23;31(12):4593-606. doi: 10.1523/JNEUROSCI.6398-10.2011. J Neurosci. 2011. PMID: 21430159 Free PMC article.
-
Calcium-Sensing Receptor Mediates β-Amyloid-Induced Synaptic Formation Impairment and Cognitive Deficits via Regulation of Cytosolic Phospholipase A2/Prostaglandin E2 Metabolic Pathway.Front Aging Neurosci. 2020 Jun 24;12:144. doi: 10.3389/fnagi.2020.00144. eCollection 2020. Front Aging Neurosci. 2020. PMID: 32670047 Free PMC article.
References
-
- Neher E, Sakaba T. Multiple roles of calcium ions in the regulation of neurotransmitter release. Neuron. 2008;59:861–872. - PubMed
-
- Geiger JR, Jonas P. Dynamic control of presynaptic Ca(2+) inflow by fast-inactivating K(+) channels in hippocampal mossy fiber boutons. Neuron. 2000;28:927–939. - PubMed
-
- Engel D, Jonas P. Presynaptic Action Potential Amplification by Voltage-Gated Na+ Channels in Hippocampal Mossy Fiber Boutons. Neuron. 2005;45:405–417. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

