A strategy for eliciting antibodies against cryptic, conserved, conformationally dependent epitopes of HIV envelope glycoprotein
- PMID: 20052405
- PMCID: PMC2797330
- DOI: 10.1371/journal.pone.0008555
A strategy for eliciting antibodies against cryptic, conserved, conformationally dependent epitopes of HIV envelope glycoprotein
Abstract
Background: Novel strategies are needed for the elicitation of broadly neutralizing antibodies to the HIV envelope glycoprotein, gp120. Experimental evidence suggests that combinations of antibodies that are broadly neutralizing in vitro may protect against challenge with HIV in nonhuman primates, and a small number of these antibodies have been selected by repertoire sampling of B cells and by the fractionation of antiserum from some patients with prolonged disease. Yet no additional strategies for identifying conserved epitopes, eliciting antibodies to these epitopes, and determining whether these epitopes are accessible to antibodies have been successful to date. The defining of additional conserved, accessible epitopes against which one can elicit antibodies will increase the probability that some may be the targets of broadly neutralizing antibodies.
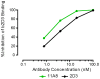
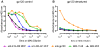
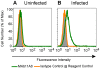
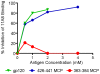
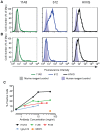
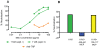
Methodology/principal findings: We postulate that additional cryptic epitopes of gp120 are present, against which neutralizing antibodies might be elicited even though these antibodies are not elicited by gp120, and that many of these epitopes may be accessible to antibodies should they be formed. We demonstrate a strategy for eliciting antibodies in mice against selected cryptic, conformationally dependent conserved epitopes of gp120 by immunizing with multiple identical copies of covalently linked peptides (MCPs). This has been achieved with MCPs representing 3 different domains of gp120. We show that some cryptic epitopes on gp120 are accessible to the elicited antibodies, and some epitopes in the CD4 binding region are not accessible. The antibodies bind to gp120 with relatively high affinity, and bind to oligomeric gp120 on the surface of infected cells.
Conclusions/significance: Immunization with MCPs comprised of selected peptides of HIV gp120 is able to elicit antibodies against conserved, conformationally dependent epitopes of gp120 that are not immunogenic when presented as gp120. Some of these cryptic epitopes are accessible to the elicited antibodies.
Conflict of interest statement
Figures

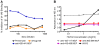


Similar articles
-
Immunogenicity and ability of variable loop-deleted human immunodeficiency virus type 1 envelope glycoproteins to elicit neutralizing antibodies.Virology. 2003 Jan 5;305(1):124-37. doi: 10.1006/viro.2002.1727. Virology. 2003. PMID: 12504547
-
Immunization with a soluble CD4-gp120 complex preferentially induces neutralizing anti-human immunodeficiency virus type 1 antibodies directed to conformation-dependent epitopes of gp120.J Virol. 1994 Sep;68(9):5854-62. doi: 10.1128/JVI.68.9.5854-5862.1994. J Virol. 1994. PMID: 7520095 Free PMC article.
-
The effect of low-profile serine substitutions in the V3 loop of HIV-1 gp120 IIIB/LAI on the immunogenicity of the envelope protein.Virology. 1998 Nov 10;251(1):59-70. doi: 10.1006/viro.1998.9392. Virology. 1998. PMID: 9813203
-
GP120: target for neutralizing HIV-1 antibodies.Annu Rev Immunol. 2006;24:739-69. doi: 10.1146/annurev.immunol.24.021605.090557. Annu Rev Immunol. 2006. PMID: 16551265 Review.
-
Antibody vs. HIV in a clash of evolutionary titans.Proc Natl Acad Sci U S A. 2005 Oct 18;102(42):14943-8. doi: 10.1073/pnas.0505126102. Epub 2005 Oct 11. Proc Natl Acad Sci U S A. 2005. PMID: 16219699 Free PMC article. Review.
Cited by
-
Deficient synthesis of class-switched, HIV-neutralizing antibodies to the CD4 binding site and correction by electrophilic gp120 immunogen.AIDS. 2014 Sep 24;28(15):2201-11. doi: 10.1097/QAD.0000000000000392. AIDS. 2014. PMID: 25022597 Free PMC article.
-
An optimally constrained V3 peptide is a better immunogen than its linear homolog or HIV-1 gp120.Virology. 2010 Jun 5;401(2):293-304. doi: 10.1016/j.virol.2010.03.007. Epub 2010 Mar 26. Virology. 2010. PMID: 20347111 Free PMC article.
-
Structural analysis of a novel rabbit monoclonal antibody R53 targeting an epitope in HIV-1 gp120 C4 region critical for receptor and co-receptor binding.Emerg Microbes Infect. 2015 Jul;4(7):e44. doi: 10.1038/emi.2015.44. Epub 2015 Jul 15. Emerg Microbes Infect. 2015. PMID: 26251831 Free PMC article.
-
A Plant-Derived Multi-HIV Antigen Induces Broad Immune Responses in Orally Immunized Mice.Mol Biotechnol. 2015 Jul;57(7):662-74. doi: 10.1007/s12033-015-9856-3. Mol Biotechnol. 2015. PMID: 25779638
-
Back to the future: covalent epitope-based HIV vaccine development.Expert Rev Vaccines. 2010 Sep;9(9):1027-43. doi: 10.1586/erv.10.77. Expert Rev Vaccines. 2010. PMID: 20822346 Free PMC article. Review.
References
-
- Wei X, Decker J, Wang S, Hui H, Kappes J, et al. Antibody neutralization and escape by HIV-1. Nature. 2003;422:307–312. - PubMed
-
- Albert J, Abrahamsson B, Nagy K, Aurelius E, Gaines H, et al. Rapid development of isolate-specific neutralizing antibodies after primary HIV-1 infection and consequent emergence of virus variants which resist neutralization by autologous sera. AIDS. 1990;4:107–112. - PubMed
-
- Arendrup M, Nielsen C, Hansen J, Mathiesen L, Nielsen J. Autologous HIV-1 neutralizing antibodies: emergence of neutralization-resistant escape virus and subsequent development of escape virus neutralizing antibodies. JAIDS Journal of Acquired Immune Deficiency Syndromes. 1992;5:303–307. - PubMed
-
- Bradney A, Scheer S, Crawford J, Buchbinder S, Montefiori D. Neutralization escape in human immunodeficiency virus type 1-infected long-term nonprogressors. The Journal of infectious diseases. 1999;179:1264–1267. - PubMed
-
- Stamatatos L, Morris L, Burton DR, Mascola JR. Neutralizing antibodies generated during natural HIV-1 infection: good news for an HIV-1 vaccine? Nat Med. 2009;15:866–870. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

