Pkd1 transgenic mice: adult model of polycystic kidney disease with extrarenal and renal phenotypes
- PMID: 20053665
- PMCID: PMC4081642
- DOI: 10.1093/hmg/ddp588
Pkd1 transgenic mice: adult model of polycystic kidney disease with extrarenal and renal phenotypes
Abstract
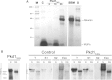
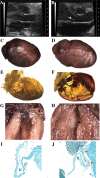
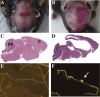
While high levels of Pkd1 expression are detected in tissues of patients with autosomal dominant polycystic kidney disease (ADPKD), it is unclear whether enhanced expression could be a pathogenetic mechanism for this systemic disorder. Three transgenic mouse lines were generated from a Pkd1-BAC modified by introducing a silent tag via homologous recombination to target a sustained wild-type genomic Pkd1 expression within the native tissue and temporal regulation. These mice specifically overexpressed the Pkd1 transgene in extrarenal and renal tissues from approximately 2- to 15-fold over Pkd1 endogenous levels in a copy-dependent manner. All transgenic mice reproducibly developed tubular and glomerular cysts leading to renal insufficiency. Interestingly, Pkd1(TAG) mice also exhibited renal fibrosis and calcium deposits in papilla reminiscent of nephrolithiasis as frequently observed in ADPKD. Similar to human ADPKD, these mice consistently displayed hepatic fibrosis and approximately 15% intrahepatic cysts of the bile ducts affecting females preferentially. Moreover, a significant proportion of mice developed cardiac anomalies with severe left-ventricular hypertrophy, marked aortic arch distention and/or valvular stenosis and calcification that had profound functional impact. Of significance, Pkd1(TAG) mice displayed occasional cerebral lesions with evidence of ruptured and unruptured cerebral aneurysms. This Pkd1(TAG) mouse model demonstrates that overexpression of wild-type Pkd1 can trigger the typical adult renal and extrarenal phenotypes resembling human ADPKD.
Figures




References
-
- Chapman A.B., Rubinstein M.D., Hughes R., Stears J.C., Earnest M.P., Johnson A.M., Gabow P.A., Kaehny W.D. Intracranial aneurysms in autosomal dominant polycystic kidney disease. N. Engl. J. Med. 1992;327:916–920. - PubMed
-
- Wei W., Hackmann K., Xu H., Germino G., Qian F. Characterization of cis-autoproteolysis of polycystin-1, the product of human polycystic kidney disease 1 gene. J. Biol. Chem. 2007;282:21729–21737. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

