Classical and/or alternative NF-kappaB pathway activation in multiple myeloma
- PMID: 20053756
- PMCID: PMC2867265
- DOI: 10.1182/blood-2009-09-243535
Classical and/or alternative NF-kappaB pathway activation in multiple myeloma
Abstract
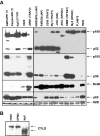
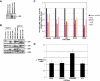
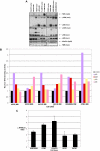
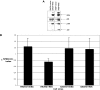
Mutations involving the nuclear factor-kappaB (NF-kappaB) pathway are present in at least 17% of multiple myeloma (MM) tumors and 40% of MM cell lines (MMCLs). These mutations, which are apparent progression events, enable MM tumors to become less dependent on bone marrow signals that activate NF-kappaB. Studies on a panel of 51 MMCLs provide some clarification of the mechanisms through which these mutations act and the significance of classical versus alternative activation of NF-kappaB. First, only one mutation (NFKB2) selectively activates the alternative pathway, whereas several mutations (CYLD, NFKB1, and TACI) selectively activate the classical pathway. However, most mutations affecting NF-kappaB-inducing kinase (NIK) levels (NIK, TRAF2, TRAF3, cIAP1&2, and CD40) activate the alternative but often both pathways. Second, we confirm the critical role of TRAF2 in regulating NIK degradation, whereas TRAF3 enhances but is not essential for cIAP1/2-mediated proteasomal degradation of NIK in MM. Third, using transfection to selectively activate the classical or alternative NF-kappaB pathways, we show virtually identical changes in gene expression in one MMCL, whereas the changes are similar albeit nonidentical in a second MMCL. Our results suggest that MM tumors can achieve increased autonomy from the bone marrow microenvironment by mutations that activate either NF-kappaB pathway.
Figures

Comment in
-
Positioning NK-kappaB in multiple myeloma.Blood. 2010 Apr 29;115(17):3422-4. doi: 10.1182/blood-2010-01-264796. Blood. 2010. PMID: 20430962 Free PMC article. No abstract available.
Similar articles
-
Promiscuous mutations activate the noncanonical NF-kappaB pathway in multiple myeloma.Cancer Cell. 2007 Aug;12(2):131-44. doi: 10.1016/j.ccr.2007.07.003. Cancer Cell. 2007. PMID: 17692805 Free PMC article.
-
Frequent engagement of the classical and alternative NF-kappaB pathways by diverse genetic abnormalities in multiple myeloma.Cancer Cell. 2007 Aug;12(2):115-30. doi: 10.1016/j.ccr.2007.07.004. Cancer Cell. 2007. PMID: 17692804 Free PMC article.
-
TACI induces cIAP1-mediated ubiquitination of NIK by TRAF2 and TANK to limit non-canonical NF-kappaB signaling.J Recept Signal Transduct Res. 2010 Apr;30(2):121-32. doi: 10.3109/10799891003634509. J Recept Signal Transduct Res. 2010. PMID: 20184394
-
Targeting signaling factors for degradation, an emerging mechanism for TRAF functions.Immunol Rev. 2015 Jul;266(1):56-71. doi: 10.1111/imr.12311. Immunol Rev. 2015. PMID: 26085207 Free PMC article. Review.
-
The noncanonical NF-κB pathway.Immunol Rev. 2012 Mar;246(1):125-40. doi: 10.1111/j.1600-065X.2011.01088.x. Immunol Rev. 2012. PMID: 22435551 Free PMC article. Review.
Cited by
-
Multi-omics tumor profiling technologies to develop precision medicine in multiple myeloma.Explor Target Antitumor Ther. 2021;2(1):65-106. doi: 10.37349/etat.2021.00034. Epub 2021 Feb 28. Explor Target Antitumor Ther. 2021. PMID: 36046090 Free PMC article. Review.
-
HMGB1 enhances chemotherapy resistance in multiple myeloma cells by activating the nuclear factor-κB pathway.Exp Ther Med. 2021 Jul;22(1):705. doi: 10.3892/etm.2021.10137. Epub 2021 May 2. Exp Ther Med. 2021. PMID: 34007314 Free PMC article.
-
Frequent upregulation of G9a promotes RelB-dependent proliferation and survival in multiple myeloma.Exp Hematol Oncol. 2020 May 22;9:8. doi: 10.1186/s40164-020-00164-4. eCollection 2020. Exp Hematol Oncol. 2020. PMID: 32477831 Free PMC article.
-
Identification of key candidate genes and pathways in multiple myeloma by integrated bioinformatics analysis.J Cell Physiol. 2019 Dec;234(12):23785-23797. doi: 10.1002/jcp.28947. Epub 2019 Jun 18. J Cell Physiol. 2019. PMID: 31215027 Free PMC article.
-
A high-risk signature for patients with multiple myeloma established from the molecular classification of human myeloma cell lines.Haematologica. 2011 Apr;96(4):574-82. doi: 10.3324/haematol.2010.033456. Epub 2010 Dec 20. Haematologica. 2011. PMID: 21173094 Free PMC article.
References
-
- Tai YT, Li XF, Breitkreutz I, et al. Role of B-cell-activating factor in adhesion and growth of human multiple myeloma cells in the bone marrow microenvironment. Cancer Res. 2006;66(13):6675–6682. - PubMed
-
- Ghosh S, Karin M. Missing pieces in the NF-kappaB puzzle. Cell. 2002;109(suppl):S81–S96. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

