Replicative senescence-associated gene expression changes in mesenchymal stromal cells are similar under different culture conditions
- PMID: 20053868
- PMCID: PMC2878782
- DOI: 10.3324/haematol.2009.011692
Replicative senescence-associated gene expression changes in mesenchymal stromal cells are similar under different culture conditions
Abstract
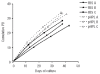
Background: Research on mesenchymal stromal cells has created high expectations for a variety of therapeutic applications. Extensive propagation to yield enough mesenchymal stromal cells for therapy may result in replicative senescence and thus hamper long-term functionality in vivo. Highly variable proliferation rates of mesenchymal stromal cells in the course of long-term expansions under varying culture conditions may already indicate different propensity for cellular senescence. We hypothesized that senescence-associated regulated genes differ in mesenchymal stromal cells propagated under different culture conditions.
Design and methods: Human bone marrow-derived mesenchymal stromal cells were cultured either by serial passaging or by a two-step protocol in three different growth conditions. Culture media were supplemented with either fetal bovine serum in varying concentrations or pooled human platelet lysate.
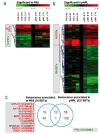
Results: All mesenchymal stromal cell preparations revealed significant gene expression changes upon long-term culture. Especially genes involved in cell differentiation, apoptosis and cell death were up-regulated, whereas genes involved in mitosis and proliferation were down-regulated. Furthermore, overlapping senescence-associated gene expression changes were found in all mesenchymal stromal cell preparations.
Conclusions: Long-term cell growth induced similar gene expression changes in mesenchymal stromal cells independently of isolation and expansion conditions. In advance of therapeutic application, this panel of genes might offer a feasible approach to assessing mesenchymal stromal cell quality with regard to the state of replicative senescence.
Figures

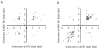

References
-
- Horwitz EM, Le BK, Dominici M, Mueller I, Slaper-Cortenbach I, Marini FC, et al. Clarification of the nomenclature for MSC: The International Society for Cellular Therapy position statement. Cytotherapy. 2005;7(5):393–5. - PubMed
-
- Stamm C, Liebold A, Steinhoff G, Strunk D. Stem cell therapy for ischemic heart disease: beginning or end of the road? Cell Transplant. 2006;15 (Suppl 1):S47–S56. - PubMed
-
- Horwitz EM, Prockop DJ, Fitzpatrick LA, Koo WW, Gordon PL, Neel M, et al. Transplantability and therapeutic effects of bone marrow-derived mesenchymal cells in children with osteogenesis imperfecta. Nat Med. 1999;5(3):309–13. - PubMed
-
- Le Blanc K, Frassoni F, Ball L, Locatelli F, Roelofs H, Lewis I, et al. Mesenchymal stem cells for treatment of steroid-resistant, severe, acute graft-versus-host disease: a phase II study. Lancet. 2008;371(9624):1579–86. - PubMed
-
- Wagner W, Ho AD. Mesenchymal stem cell preparations–comparing apples and oranges. Stem Cell Rev. 2007;3(4):239–48. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

