Reducing intracortical inhibition in the adult visual cortex promotes ocular dominance plasticity
- PMID: 20053917
- PMCID: PMC6632513
- DOI: 10.1523/JNEUROSCI.2233-09.2010
Reducing intracortical inhibition in the adult visual cortex promotes ocular dominance plasticity
Abstract
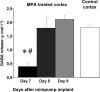
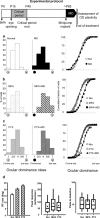
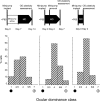
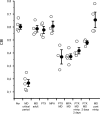
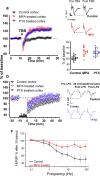
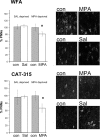
Experience-dependent plasticity in the cortex is often higher during short critical periods in postnatal development. The mechanisms limiting adult cortical plasticity are still unclear. Maturation of intracortical GABAergic inhibition is suggested to be crucial for the closure of the critical period for ocular dominance (OD) plasticity in the visual cortex. We find that reduction of GABAergic transmission in the adult rat visual cortex partially reactivates OD plasticity in response to monocular deprivation (MD). This is accompanied by an enhancement of activity-dependent potentiation of synaptic efficacy but not of activity-dependent depression. We also found a decrease in the expression of chondroitin sulfate proteoglycans in the visual cortex of MD animals with reduced inhibition, after the reactivation of OD plasticity. Thus, intracortical inhibition is a crucial limiting factor for the induction of experience-dependent plasticity in the adult visual cortex.
Figures
Similar articles
-
Experience-dependent reactivation of ocular dominance plasticity in the adult visual cortex.Exp Neurol. 2010 Nov;226(1):100-9. doi: 10.1016/j.expneurol.2010.08.009. Epub 2010 Aug 14. Exp Neurol. 2010. PMID: 20713044
-
A rich environmental experience reactivates visual cortex plasticity in aged rats.Exp Gerontol. 2012 Apr;47(4):337-41. doi: 10.1016/j.exger.2012.01.007. Epub 2012 Feb 4. Exp Gerontol. 2012. PMID: 22329907
-
Visual deprivation reactivates rapid ocular dominance plasticity in adult visual cortex.J Neurosci. 2006 Mar 15;26(11):2951-5. doi: 10.1523/JNEUROSCI.5554-05.2006. J Neurosci. 2006. PMID: 16540572 Free PMC article.
-
Lifelong learning: ocular dominance plasticity in mouse visual cortex.Curr Opin Neurobiol. 2006 Aug;16(4):451-9. doi: 10.1016/j.conb.2006.06.007. Epub 2006 Jul 11. Curr Opin Neurobiol. 2006. PMID: 16837188 Review.
-
Plasticity in the adult brain: lessons from the visual system.Exp Brain Res. 2009 Jan;192(3):335-41. doi: 10.1007/s00221-008-1509-3. Epub 2008 Jul 31. Exp Brain Res. 2009. PMID: 18668231 Review.
Cited by
-
Activity-dependent plasticity and gene expression modifications in the adult CNS.Front Mol Neurosci. 2011 Nov 28;4:50. doi: 10.3389/fnmol.2011.00050. eCollection 2011. Front Mol Neurosci. 2011. PMID: 22144945 Free PMC article.
-
Visual system plasticity in mammals: the story of monocular enucleation-induced vision loss.Front Syst Neurosci. 2015 Apr 28;9:60. doi: 10.3389/fnsys.2015.00060. eCollection 2015. Front Syst Neurosci. 2015. PMID: 25972788 Free PMC article. Review.
-
GABAergic circuit dysfunctions in neurodevelopmental disorders.Front Psychiatry. 2012 May 31;3:51. doi: 10.3389/fpsyt.2012.00051. eCollection 2012. Front Psychiatry. 2012. PMID: 22666213 Free PMC article.
-
Candidate genes in ocular dominance plasticity.Front Neurosci. 2012 Feb 1;6:11. doi: 10.3389/fnins.2012.00011. eCollection 2012. Front Neurosci. 2012. PMID: 22347157 Free PMC article.
-
Synapse-Selective Control of Cortical Maturation and Plasticity by Parvalbumin-Autonomous Action of SynCAM 1.Cell Rep. 2019 Jan 8;26(2):381-393.e6. doi: 10.1016/j.celrep.2018.12.069. Cell Rep. 2019. PMID: 30625321 Free PMC article.
References
-
- Baranes D, Lederfein D, Huang YY, Chen M, Bailey CH, Kandel ER. Tissue plasminogen activator contributes to the late phase of LTP and to synaptic growth in the hippocampal mossy fiber pathway. Neuron. 1998;21:813–825. - PubMed
-
- Benevento LA, Bakkum BW, Cohen RS. gamma-Aminobutyric acid and somatostatin immunoreactivity in the visual cortex of normal and dark-reared rats. Brain Res. 1995;689:172–182. - PubMed
-
- Berardi N, Pizzorusso T, Maffei L. Critical periods during sensory development. Curr Opin Neurobiol. 2000;10:138–145. - PubMed
-
- Berardi N, Pizzorusso T, Ratto GM, Maffei L. Molecular basis of plasticity in the visual cortex. Trends Neurosci. 2003;26:369–378. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
