Wild-type cone photoreceptors persist despite neighboring mutant cone degeneration
- PMID: 20053919
- PMCID: PMC2805452
- DOI: 10.1523/JNEUROSCI.5019-09.2010
Wild-type cone photoreceptors persist despite neighboring mutant cone degeneration
Abstract
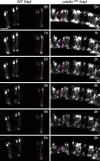
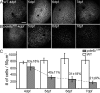
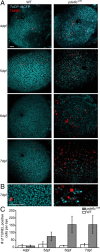
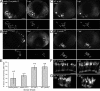
In many retinal diseases, the malfunction that results in photoreceptor loss occurs only in either rods or cones, but degeneration can progress from the affected cell type to its healthy neighbors. Specifically, in human and mouse models of Retinitis Pigmentosa the loss of rods results in the death of neighboring healthy cones. Significantly less is known about cone-initiated degenerations and their affect on neighboring cells. Sometimes rods remain normal after cone death, whereas other patients experience a loss of scotopic vision over time. The affect of cone death on neighboring cones is unknown. The zebrafish is a cone-rich animal model in which the potential for dying cones to kill neighboring healthy cones can be evaluated. We previously reported that the zebrafish cone phosphodiesterase mutant (pde6c(w59)) displays a rapid death of cones soon after their formation and a subsequent loss of rods in the central retina. In this study we examine morphological changes associated with cone death in vivo in pde6c(w59) fish. We then use blastulae transplantations to create chimeric fish with a photoreceptor layer of mixed wild-type (WT) and pde6c(w59) cones. We find that the death of inoperative cones does not cause neighboring WT cone loss. The survival of WT cones is independent of transplant size and location within the retina. Furthermore, transplanted WT cones persist at least several weeks after the initial death of dysfunctional mutant cones. Our results suggest a potential for the therapeutic transplantation of healthy cones into an environment of damaged cones.
Figures


Similar articles
-
Rip3 knockdown rescues photoreceptor cell death in blind pde6c zebrafish.Cell Death Differ. 2014 May;21(5):665-75. doi: 10.1038/cdd.2013.191. Epub 2014 Jan 10. Cell Death Differ. 2014. PMID: 24413151 Free PMC article.
-
Rod photoreceptors protect from cone degeneration-induced retinal remodeling and restore visual responses in zebrafish.J Neurosci. 2013 Jan 30;33(5):1804-14. doi: 10.1523/JNEUROSCI.2910-12.2013. J Neurosci. 2013. PMID: 23365220 Free PMC article.
-
Robust cone-mediated signaling persists late into rod photoreceptor degeneration.Elife. 2022 Aug 30;11:e80271. doi: 10.7554/eLife.80271. Elife. 2022. PMID: 36040015 Free PMC article.
-
Therapeutic strategy for handling inherited retinal degenerations in a gene-independent manner using rod-derived cone viability factors.C R Biol. 2014 Mar;337(3):207-13. doi: 10.1016/j.crvi.2013.12.002. Epub 2014 Feb 20. C R Biol. 2014. PMID: 24702847 Review.
-
A review of the mechanisms of cone degeneration in retinitis pigmentosa.Acta Ophthalmol. 2016 Dec;94(8):748-754. doi: 10.1111/aos.13141. Epub 2016 Jun 27. Acta Ophthalmol. 2016. PMID: 27350263 Review.
Cited by
-
Mutant Prpf31 causes pre-mRNA splicing defects and rod photoreceptor cell degeneration in a zebrafish model for Retinitis pigmentosa.Mol Neurodegener. 2011 Jul 30;6:56. doi: 10.1186/1750-1326-6-56. Mol Neurodegener. 2011. PMID: 21801444 Free PMC article.
-
Loss of Pde6 reduces cell body Ca(2+) transients within photoreceptors.Cell Death Dis. 2013 Sep 12;4(9):e797. doi: 10.1038/cddis.2013.332. Cell Death Dis. 2013. PMID: 24030149 Free PMC article.
-
Deletion of the X-linked opsin gene array locus control region (LCR) results in disruption of the cone mosaic.Vision Res. 2010 Sep 15;50(19):1989-99. doi: 10.1016/j.visres.2010.07.009. Epub 2010 Jul 16. Vision Res. 2010. PMID: 20638402 Free PMC article.
-
A Naturally-Derived Compound Schisandrin B Enhanced Light Sensation in the pde6c Zebrafish Model of Retinal Degeneration.PLoS One. 2016 Mar 1;11(3):e0149663. doi: 10.1371/journal.pone.0149663. eCollection 2016. PLoS One. 2016. PMID: 26930483 Free PMC article.
-
Biochemical adaptations of the retina and retinal pigment epithelium support a metabolic ecosystem in the vertebrate eye.Elife. 2017 Sep 13;6:e28899. doi: 10.7554/eLife.28899. Elife. 2017. PMID: 28901286 Free PMC article.
References
-
- Andrade-Rozental AF, Rozental R, Hopperstad MG, Wu JK, Vrionis FD, Spray DC. Gap junctions: the “kiss of death” and the “kiss of life.”. Brain Res Brain Res Rev. 2000;32:308–315. - PubMed
-
- Branchek T. The development of photoreceptors in the zebrafish, brachydanio rerio. II. Function. J Comp Neurol. 1984;224:116–122. - PubMed
-
- Brockerhoff SE. Measuring the optokinetic response of zebrafish larvae. Nat Protoc. 2006;1:2448–2451. - PubMed
-
- Carmany-Rampey A, Moens CB. Modern mosaic analysis in the zebrafish. Methods. 2006;39:228–238. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
