Molecular and biochemical characterization of folate transport proteins in retinal Müller cells
- PMID: 20053979
- PMCID: PMC2891475
- DOI: 10.1167/iovs.09-4833
Molecular and biochemical characterization of folate transport proteins in retinal Müller cells
Abstract
Purpose: To analyze the mechanisms of folate uptake in retinal Müller cells.
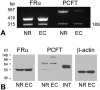
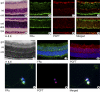
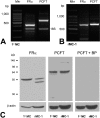
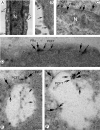
Methods: RT-PCR and Western blot analysis were performed in freshly isolated neural retina and RPE/eyecup, primary mouse Müller cells, and rMC-1 cells for the three known folate transport proteins folate receptor alpha (FRalpha), proton-coupled folate transporter (PCFT), and reduced folate carrier (RFC). Laser scanning confocal microscopy (LSCM) and immunoelectron microscopy were used to determine the subcellular location of FRalpha and PCFT in primary Müller cells. The pH dependence of the uptake of [(3)H]-methyltetrahydrofolate ([(3)H]-MTF) was assayed in Müller cells in the presence/absence of thiamine pyrophosphate, an inhibitor of RFC.
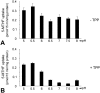
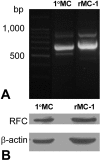
Results: FRalpha and PCFT are expressed abundantly in the retina in several cell layers, including the inner nuclear layer; they are present in primary mouse Müller cells and rMC-1 cells. LSCM localized these proteins to the plasma membrane, nuclear membrane, and perinuclear region. Immunoelectron microscopic studies revealed the colocalization of FRalpha and PCFT on the plasma membrane and nuclear membrane and within endosomal structures. Müller cell uptake of [(3)H]-MTF was robust at pH 5.0 to 6.0, consistent with PCFT activity, but also at neutral pH, reflecting RFC function. RFC was expressed in mouse Müller cells that had been allowed to proliferate in culture, but not in freshly isolated primary cells.
Conclusions: FRalpha and PCFT are expressed in retinal Müller cells and colocalize in the endosomal compartment, suggesting that the two proteins may work coordinately to mediate folate uptake. The unexpected finding of RFC expression and activity in cultured Müller cells may reflect the upregulation of this protein under proliferative conditions.
Figures


References
-
- Knox DL, Chen MF, Guilarte TR, Dang CV, Burnette J. Nutritional amblyopia: folic acid, vitamin B-12, and other vitamins. Retina 1982;2:288–293 - PubMed
-
- Smiddy WE, Green WR. Nutritional amblyopia: a histopathologic study with retrospective clinical correlation. Graefe's Arch Clin Exp Ophthalmol 1987;225:321–324 - PubMed
-
- Golnik KC, Schaible ER. Folate-responsive optic neuropathy. J Neurophthalmol 1994;14:163–169 - PubMed
-
- Miller NR. The optic nerve. Curr Opin Neurol 1996;9:5–15 - PubMed
-
- Sadun A, Rubin R. Residual psychophysical deficits following recovery from the Cuban epidemic of optic neuropathy. In: Lakshminarayan V. ed. Basic and Clinical Applications of Vision Science Dordrecht: Kluwer; 1997:231–234
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

