Sucrose octasulfate selectively accelerates thrombin inactivation by heparin cofactor II
- PMID: 20053992
- PMCID: PMC2832979
- DOI: 10.1074/jbc.M109.005967
Sucrose octasulfate selectively accelerates thrombin inactivation by heparin cofactor II
Abstract
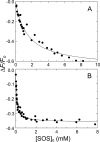
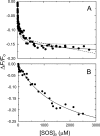
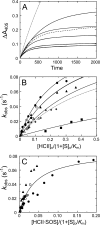
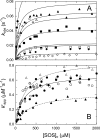
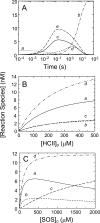
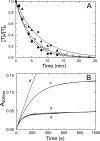
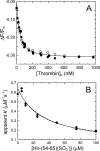
Inactivation of thrombin (T) by the serpins heparin cofactor II (HCII) and antithrombin (AT) is accelerated by a heparin template between the serpin and thrombin exosite II. Unlike AT, HCII also uses an allosteric interaction of its NH(2)-terminal segment with exosite I. Sucrose octasulfate (SOS) accelerated thrombin inactivation by HCII but not AT by 2000-fold. SOS bound to two sites on thrombin, with dissociation constants (K(D)) of 10 +/- 4 microm and 400 +/- 300 microm that were not kinetically resolvable, as evidenced by single hyperbolic SOS concentration dependences of the inactivation rate (k(obs)). SOS bound HCII with K(D) 1.45 +/- 0.30 mm, and this binding was tightened in the T.SOS.HCII complex, characterized by K(complex) of approximately 0.20 microm. Inactivation data were incompatible with a model solely depending on HCII.SOS but fit an equilibrium linkage model employing T.SOS binding in the pathway to higher order complex formation. Hirudin-(54-65)(SO(3)(-)) caused a hyperbolic decrease of the inactivation rates, suggesting partial competitive binding of hirudin-(54-65)(SO(3)(-)) and HCII to exosite I. Meizothrombin(des-fragment 1), binding SOS with K(D) = 1600 +/- 300 microm, and thrombin were inactivated at comparable rates, and an exosite II aptamer had no effect on the inactivation, suggesting limited exosite II involvement. SOS accelerated inactivation of meizothrombin 1000-fold, reflecting the contribution of direct exosite I interaction with HCII. Thrombin generation in plasma was suppressed by SOS, both in HCII-dependent and -independent processes. The ex vivo HCII-dependent process may utilize the proposed model and suggests a potential for oversulfated disaccharides in controlling HCII-regulated thrombin generation.
Figures

References
-
- Olson S. T. (1988) J. Biol. Chem. 263, 1698–1708 - PubMed
-
- Tollefsen D. M., Pestka C. A., Monafo W. J. (1983) J. Biol. Chem. 258, 6713–6716 - PubMed
-
- Van Deerlin V. M., Tollefsen D. M. (1991) J. Biol. Chem. 266, 20223–20231 - PubMed
-
- Rogers S. J., Pratt C. W., Whinna H. C., Church F. C. (1992) J. Biol. Chem. 267, 3613–3617 - PubMed
-
- Church F. C., Meade J. B., Treanor R. E., Whinna H. C. (1989) J. Biol. Chem. 264, 3618–3623 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

