A novel mannose-binding lectin/ficolin-associated protein is highly expressed in heart and skeletal muscle tissues and inhibits complement activation
- PMID: 20053996
- PMCID: PMC2832975
- DOI: 10.1074/jbc.M109.065805
A novel mannose-binding lectin/ficolin-associated protein is highly expressed in heart and skeletal muscle tissues and inhibits complement activation
Abstract
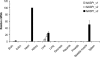
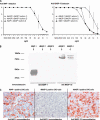
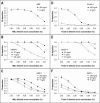
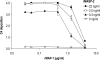
The human lectin complement pathway involves circulating complexes consisting of mannose-binding lectin (MBL) or three ficolins (ficolin-1, -2, and -3) in association with three MBL/ficolin-associated serine proteases (MASP) (MASP-1, -2, and -3) and a nonenzymatic sMAP. MASP-1 and MASP-3 (MASP1 isoforms 1 and 2, respectively) are splice variants of the MASP1 gene, whereas MASP-2 and sMAP are splice variants of the MASP2 gene. We have identified a novel serum protein of 45 kDa that is associated with MBL and the ficolins. This protein is named MBL/ficolin-associated protein 1 (MAP-1 corresponding to MASP1 isoform 3). The transcript generating MAP-1 (MASP1_v3) contains exons 1-8 and a novel exon encoding an in-frame stop codon. The corresponding protein lacks the serine protease domains but contains most of the common heavy chain of MASP-1 and MASP-3. Additionally MAP-1 contains 17 unique C-terminal amino acids. By use of quantitative PCR and MAP-1-specific immunohistochemistry, we found that MAP-1 is highly expressed in myocardial and skeletal muscle tissues as well as in liver hepatocytes with a different expression profile than that observed for MASP-1 and MASP-3. MAP-1 co-precipitated from human serum with MBL, ficolin-2, and ficolin-3, and recombinant MAP-1 was able to inhibit complement C4 deposition via both the ficolin-3 and MBL pathway. In conclusion we have identified a novel 45-kDa serum protein derived from the MASP1 gene, which is highly expressed in striated muscle tissues. It is found in complex with MBL and ficolins and may function as a potent inhibitor of the complement system in vivo.
Figures




Similar articles
-
Localization and characterization of the mannose-binding lectin (MBL)-associated-serine protease-2 binding site in rat ficolin-A: equivalent binding sites within the collagenous domains of MBLs and ficolins.J Immunol. 2007 Jul 1;179(1):455-62. doi: 10.4049/jimmunol.179.1.455. J Immunol. 2007. PMID: 17579066 Free PMC article.
-
Crystal structure and functional characterization of the complement regulator mannose-binding lectin (MBL)/ficolin-associated protein-1 (MAP-1).J Biol Chem. 2012 Sep 21;287(39):32913-21. doi: 10.1074/jbc.M112.386680. Epub 2012 Aug 1. J Biol Chem. 2012. PMID: 22854970 Free PMC article.
-
Heterocomplex formation between MBL/ficolin/CL-11-associated serine protease-1 and -3 and MBL/ficolin/CL-11-associated protein-1.J Immunol. 2014 May 1;192(9):4352-60. doi: 10.4049/jimmunol.1303263. Epub 2014 Mar 28. J Immunol. 2014. PMID: 24683193
-
Structural and functional overview of the lectin complement pathway: its molecular basis and physiological implication.Arch Immunol Ther Exp (Warsz). 2013 Aug;61(4):273-83. doi: 10.1007/s00005-013-0229-y. Epub 2013 Apr 7. Arch Immunol Ther Exp (Warsz). 2013. PMID: 23563865 Review.
-
Role of MBL-associated serine protease (MASP) on activation of the lectin complement pathway.Adv Exp Med Biol. 2007;598:93-104. doi: 10.1007/978-0-387-71767-8_8. Adv Exp Med Biol. 2007. PMID: 17892207 Review.
Cited by
-
MBL-associated serine proteases (MASPs) and infectious diseases.Mol Immunol. 2015 Sep;67(1):85-100. doi: 10.1016/j.molimm.2015.03.245. Epub 2015 Apr 8. Mol Immunol. 2015. PMID: 25862418 Free PMC article. Review.
-
Protection of host cells by complement regulators.Immunol Rev. 2016 Nov;274(1):152-171. doi: 10.1111/imr.12475. Immunol Rev. 2016. PMID: 27782321 Free PMC article. Review.
-
CL-L1 and CL-K1 Exhibit Widespread Tissue Distribution With High and Co-Localized Expression in Secretory Epithelia and Mucosa.Front Immunol. 2018 Jul 31;9:1757. doi: 10.3389/fimmu.2018.01757. eCollection 2018. Front Immunol. 2018. PMID: 30108587 Free PMC article.
-
The emerging role of complement lectin pathway in trypanosomatids: molecular bases in activation, genetic deficiencies, susceptibility to infection, and complement system-based therapeutics.ScientificWorldJournal. 2013;2013:675898. doi: 10.1155/2013/675898. Epub 2013 Feb 21. ScientificWorldJournal. 2013. PMID: 23533355 Free PMC article. Review.
-
Factors involved in initiation and regulation of complement lectin pathway influence postoperative outcome after pediatric cardiac surgery involving cardiopulmonary bypass.Sci Rep. 2019 Feb 27;9(1):2930. doi: 10.1038/s41598-019-39742-w. Sci Rep. 2019. PMID: 30814659 Free PMC article.
References
-
- Walport M. J. (2001) N. Engl. J. Med. 344, 1058–1066 - PubMed
-
- Endo Y., Matsushita M., Fujita T. (2007) Immunobiology 212, 371–379 - PubMed
-
- Turner M. W. (2003) Mol. Immunol. 40, 423–429 - PubMed
-
- Fujita T. (2002) Nat. Rev. Immunol. 2, 346–353 - PubMed
-
- Thiel S., Vorup-Jensen T., Stover C. M., Schwaeble W., Laursen S. B., Poulsen K., Willis A. C., Eggleton P., Hansen S., Holmskov U., Reid K. B., Jensenius J. C. (1997) Nature 386, 506–510 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

