Novel peptoid building blocks: synthesis of functionalized aromatic helix-inducing submonomers
- PMID: 20055478
- PMCID: PMC2814525
- DOI: 10.1021/ol902660p
Novel peptoid building blocks: synthesis of functionalized aromatic helix-inducing submonomers
Abstract
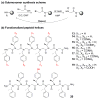
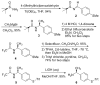
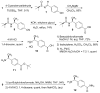
Peptoids, oligo-N-substituted glycines, can fold into well-defined helical secondary structures. The design and synthesis of new peptoid building blocks that are capable of both (a) inducing a helical secondary structure and (b) decorating the helices with chemical functionalities are reported. Peptoid heptamers containing carboxamide, carboxylic acid or thiol functionalities were synthesized, and the resulting peptoids were shown to form stable helices. A thiol-containing peptoid readily formed the homodisulfide, providing a convenient route to prepare peptoid helix homodimers.
Figures


Similar articles
-
Extraordinarily robust polyproline type I peptoid helices generated via the incorporation of α-chiral aromatic N-1-naphthylethyl side chains.J Am Chem Soc. 2011 Oct 5;133(39):15559-67. doi: 10.1021/ja204755p. Epub 2011 Sep 13. J Am Chem Soc. 2011. PMID: 21861531 Free PMC article.
-
Synthesis and characterization of nitroaromatic peptoids: fine tuning peptoid secondary structure through monomer position and functionality.J Org Chem. 2009 Feb 20;74(4):1440-9. doi: 10.1021/jo8023363. J Org Chem. 2009. PMID: 19159244 Free PMC article.
-
Peptoid oligomers with alpha-chiral, aromatic side chains: sequence requirements for the formation of stable peptoid helices.J Am Chem Soc. 2001 Jul 18;123(28):6778-84. doi: 10.1021/ja003154n. J Am Chem Soc. 2001. PMID: 11448181
-
Peptoid architectures: elaboration, actuation, and application.Curr Opin Chem Biol. 2008 Dec;12(6):714-21. doi: 10.1016/j.cbpa.2008.08.015. Epub 2008 Sep 9. Curr Opin Chem Biol. 2008. PMID: 18786652 Review.
-
Helices in peptoids of alpha- and beta-peptides.Phys Biol. 2006 Feb 2;3(1):S1-9. doi: 10.1088/1478-3975/3/1/S01. Phys Biol. 2006. PMID: 16582460 Review.
Cited by
-
Synthesis of boron-containing primary amines.Molecules. 2013 Oct 8;18(10):12346-67. doi: 10.3390/molecules181012346. Molecules. 2013. PMID: 24108399 Free PMC article.
-
Practical and Metal-Free Synthesis of Novel Enantiopure Amides Containing the Potentially Bioactive 5-Nitroimidazole Moiety.Molecules. 2016 Nov 4;21(11):1472. doi: 10.3390/molecules21111472. Molecules. 2016. PMID: 27827934 Free PMC article.
-
Applications of Discrete Synthetic Macromolecules in Life and Materials Science: Recent and Future Trends.Adv Sci (Weinh). 2021 Jan 25;8(6):2004038. doi: 10.1002/advs.202004038. eCollection 2021 Mar. Adv Sci (Weinh). 2021. PMID: 33747749 Free PMC article. Review.
-
A rotamer library to enable modeling and design of peptoid foldamers.J Am Chem Soc. 2014 Jun 18;136(24):8772-82. doi: 10.1021/ja503776z. Epub 2014 Jun 9. J Am Chem Soc. 2014. PMID: 24823488 Free PMC article.
-
Log D versus HPLC derived hydrophobicity: The development of predictive tools to aid in the rational design of bioactive peptoids.Biopolymers. 2017 Jul;108(4):e23014. doi: 10.1002/bip.23014. Biopolymers. 2017. PMID: 28085180 Free PMC article.
References
-
- Branden C, Tooze J. Introduction to Protein Structure. 2nd. Garland Publishing Inc.; New York, NY: 1999.
-
- Barron AE, Zuckermann RN. Curr Opin Chem Biol. 1999;3:681–687. - PubMed
-
- Simons RJ, Kania RS, Zuckermann RN, Huebner VD, Jewell DA, Banville S, Ng S, Wang L, Rosenberg S, Marlowe CK, Spellmeyer DC, Tan R, Frankel AD, Santi DV, Cohen FE, Bartlett PA. Proc Natl Acad Sci USA. 1992;89:9367–9371. - PMC - PubMed
- Fowler SA, Blackwell HE. Org Biomol Chem. 2009;7:1508–1524. - PMC - PubMed
-
- Zuckermann RN, Kerr JM, Kent SBH, Moos WH. J Am Chem Soc. 1992;114:10646–10647.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

