Heat denatured enzymatically inactive recombinant chlamydial protease-like activity factor induces robust protective immunity against genital chlamydial challenge
- PMID: 20056182
- PMCID: PMC2846592
- DOI: 10.1016/j.vaccine.2009.12.064
Heat denatured enzymatically inactive recombinant chlamydial protease-like activity factor induces robust protective immunity against genital chlamydial challenge
Abstract
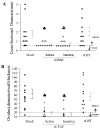
We have shown previously that vaccination with recombinant chlamydial protease-like activity factor (rCPAF) plus interleukin-12 as an adjuvant induces robust protective immunity against primary genital Chlamydia muridarum challenge in mice. Since CPAF is a protease, we compared the effects of enzymatically active and inactive (heat denatured) rCPAF to determine whether proteolytic activity is expendable for the induction of protective immunity against chlamydial challenge. Active, but not inactive, rCPAF immunization induced high levels of anti-active CPAF antibody, whereas both induced robust splenic CPAF-specific IFN-gamma production. Vaccination with active or inactive rCPAF induced enhanced vaginal chlamydial clearance as early as day 6 with complete resolution of infection by day 18, compared to day 30 in mock-vaccinated and challenged animals. Importantly, significant and comparable reductions in oviduct pathology were observed in active and inactive rCPAF-vaccinated mice compared to mock-vaccinated animals. Thus, rCPAF induced anti-chlamydial immunity is largely independent of enzymatic activity and secondary or higher order protein conformation.
Copyright 2009 Elsevier Ltd. All rights reserved.
Figures




Similar articles
-
Intranasal vaccination with a secreted chlamydial protein enhances resolution of genital Chlamydia muridarum infection, protects against oviduct pathology, and is highly dependent upon endogenous gamma interferon production.Infect Immun. 2007 Feb;75(2):666-76. doi: 10.1128/IAI.01280-06. Epub 2006 Nov 21. Infect Immun. 2007. PMID: 17118987 Free PMC article.
-
Immunization with a combination of integral chlamydial antigens and a defined secreted protein induces robust immunity against genital chlamydial challenge.Infect Immun. 2010 Sep;78(9):3942-9. doi: 10.1128/IAI.00346-10. Epub 2010 Jul 6. Infect Immun. 2010. PMID: 20605976 Free PMC article.
-
Induction of cross-serovar protection against genital chlamydial infection by a targeted multisubunit vaccination approach.Clin Vaccine Immunol. 2007 Dec;14(12):1537-44. doi: 10.1128/CVI.00274-07. Epub 2007 Oct 17. Clin Vaccine Immunol. 2007. PMID: 17942608 Free PMC article.
-
Vaccination against Chlamydia genital infection utilizing the murine C. muridarum model.Infect Immun. 2011 Mar;79(3):986-96. doi: 10.1128/IAI.00881-10. Epub 2010 Nov 15. Infect Immun. 2011. PMID: 21078844 Free PMC article. Review.
-
Chlamydial protease-like activity factor--insights into immunity and vaccine development.J Reprod Immunol. 2009 Dec;83(1-2):179-84. doi: 10.1016/j.jri.2009.05.007. Epub 2009 Oct 23. J Reprod Immunol. 2009. PMID: 19853923 Free PMC article. Review.
Cited by
-
Antibody signature of spontaneous clearance of Chlamydia trachomatis ocular infection and partial resistance against re-challenge in a nonhuman primate trachoma model.PLoS Negl Trop Dis. 2013 May 30;7(5):e2248. doi: 10.1371/journal.pntd.0002248. Print 2013. PLoS Negl Trop Dis. 2013. PMID: 23738030 Free PMC article.
-
Chlamydia muridarum T cell antigens and adjuvants that induce protective immunity in mice.Infect Immun. 2012 Apr;80(4):1510-8. doi: 10.1128/IAI.06338-11. Epub 2012 Jan 30. Infect Immun. 2012. PMID: 22290151 Free PMC article.
-
Induction of immune memory by a multisubunit chlamydial vaccine.Vaccine. 2011 Feb 4;29(7):1472-80. doi: 10.1016/j.vaccine.2010.12.024. Epub 2010 Dec 22. Vaccine. 2011. PMID: 21184858 Free PMC article.
-
Viral-vectored boosting of OmcB- or CPAF-specific T-cell responses fail to enhance protection from Chlamydia muridarum in infection-immune mice and elicits a non-protective CD8-dominant response in naïve mice.Mucosal Immunol. 2024 Oct;17(5):1005-1018. doi: 10.1016/j.mucimm.2024.06.012. Epub 2024 Jul 3. Mucosal Immunol. 2024. PMID: 38969067 Free PMC article.
-
TLR4-mediated immunomodulatory properties of the bacterial metalloprotease arazyme in preclinical tumor models.Oncoimmunology. 2016 May 5;5(7):e1178420. doi: 10.1080/2162402X.2016.1178420. eCollection 2016 Jul. Oncoimmunology. 2016. PMID: 27622031 Free PMC article.
References
-
- Brunham RC, Rey-Ladino J. Immunology of Chlamydia infection: implications for a Chlamydia trachomatis vaccine. Nat Rev Immunol. 2005 Feb;5(2):149–161. - PubMed
-
- World Health Organization. Global Prevalence and Incidence of Selected Curable Sexually Transmitted Infections: Overview and Estimates. 2001:11–15.
-
- Kelly KA. Cellular immunity and Chlamydia genital infection: induction, recruitment, and effector mechanisms. Int Rev Immunol. 2003 Jan;22(1):3–41. - PubMed
-
- Rekart ML, Brunham RC. Epidemiology of chlamydial infection: are we losing ground? Sex Transm Infect. 2008 Apr;84(2):87–91. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

