Mevalonate analogues as substrates of enzymes in the isoprenoid biosynthetic pathway of Streptococcus pneumoniae
- PMID: 20056424
- PMCID: PMC2842986
- DOI: 10.1016/j.bmc.2009.12.050
Mevalonate analogues as substrates of enzymes in the isoprenoid biosynthetic pathway of Streptococcus pneumoniae
Abstract
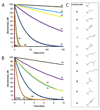
Survival of the human pathogen Streptococcus pneumoniae requires a functional mevalonate pathway, which produces isopentenyl diphosphate, the essential building block of isoprenoids. Flux through this pathway appears to be regulated at the mevalonate kinase (MK) step, which is strongly feedback-inhibited by diphosphomevalonate (DPM), the penultimate compound in the pathway. The human mevalonate pathway is not regulated by DPM, making the bacterial pathway an attractive antibiotic target. Since DPM has poor drug characteristics, being highly charged, we propose to use unphosphorylated, cell-permeable prodrugs based on mevalonate that will be phosphorylated in turn by MK and phosphomevalonate kinase (PMK) to generate the active compound in situ. To test the limits of this approach, we synthesized a series of C(3)-substituted mevalonate analogues to probe the steric and electronic requirements of the MK and PMK active sites. MK and PMK accepted substrates with up to two additional carbons, showing a preference for small substituents. This result establishes the feasibility of using a prodrug strategy for DPM-based antibiotics in S. pneumoniae and identified several analogues to be tested as inhibitors of MK. Among the substrates accepted by both enzymes were cyclopropyl, vinyl, and ethynyl mevalonate analogues that, when diphosphorylated, might be mechanism-based inactivators of the next enzyme in the pathway, diphosphomevalonate decarboxylase.
Copyright (c) 2009 Elsevier Ltd. All rights reserved.
Figures




Similar articles
-
Probing ligand-binding pockets of the mevalonate pathway enzymes from Streptococcus pneumoniae.J Biol Chem. 2010 Jul 2;285(27):20654-63. doi: 10.1074/jbc.M109.098350. Epub 2010 Apr 19. J Biol Chem. 2010. PMID: 20404339 Free PMC article.
-
Crystal structure of the Streptococcus pneumoniae mevalonate kinase in complex with diphosphomevalonate.Protein Sci. 2007 May;16(5):983-9. doi: 10.1110/ps.072755707. Epub 2007 Mar 30. Protein Sci. 2007. PMID: 17400916 Free PMC article.
-
Streptococcus pneumoniae isoprenoid biosynthesis is downregulated by diphosphomevalonate: an antimicrobial target.Biochemistry. 2004 Dec 28;43(51):16461-6. doi: 10.1021/bi048075t. Biochemistry. 2004. PMID: 15610040
-
Enzymes of the mevalonate pathway of isoprenoid biosynthesis.Arch Biochem Biophys. 2011 Jan 15;505(2):131-43. doi: 10.1016/j.abb.2010.09.028. Epub 2010 Oct 7. Arch Biochem Biophys. 2011. PMID: 20932952 Free PMC article. Review.
-
Biochemical and genetic aspects of mevalonate kinase and its deficiency.Biochim Biophys Acta. 2000 Dec 15;1529(1-3):19-32. doi: 10.1016/s1388-1981(00)00135-9. Biochim Biophys Acta. 2000. PMID: 11111075 Review.
Cited by
-
Probing ligand-binding pockets of the mevalonate pathway enzymes from Streptococcus pneumoniae.J Biol Chem. 2010 Jul 2;285(27):20654-63. doi: 10.1074/jbc.M109.098350. Epub 2010 Apr 19. J Biol Chem. 2010. PMID: 20404339 Free PMC article.
-
Synthesis of mevalonate- and fluorinated mevalonate prodrugs and their in vitro human plasma stability.Eur J Med Chem. 2015 Jan 27;90:448-61. doi: 10.1016/j.ejmech.2014.11.040. Epub 2014 Nov 22. Eur J Med Chem. 2015. PMID: 25461893 Free PMC article.
-
An Artificial Pathway for Isoprenoid Biosynthesis Decoupled from Native Hemiterpene Metabolism.ACS Synth Biol. 2019 Feb 15;8(2):232-238. doi: 10.1021/acssynbio.8b00383. Epub 2019 Jan 16. ACS Synth Biol. 2019. PMID: 30648856 Free PMC article.
-
Characterization of novel mevalonate kinases from the tardigrade Ramazzottius varieornatus and the psychrophilic archaeon Methanococcoides burtonii.Acta Crystallogr D Struct Biol. 2024 Mar 1;80(Pt 3):203-215. doi: 10.1107/S2059798324001360. Epub 2024 Feb 27. Acta Crystallogr D Struct Biol. 2024. PMID: 38411551 Free PMC article.
-
A linear pathway for mevalonate production supports growth of Thermococcus kodakarensis.Extremophiles. 2019 Mar;23(2):229-238. doi: 10.1007/s00792-019-01076-w. Epub 2019 Jan 23. Extremophiles. 2019. PMID: 30673855
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

