Fluorosomes: a convenient new reagent to detect and block multivalent and complex receptor-ligand interactions
- PMID: 20056716
- PMCID: PMC2879947
- DOI: 10.1096/fj.09-137281
Fluorosomes: a convenient new reagent to detect and block multivalent and complex receptor-ligand interactions
Abstract
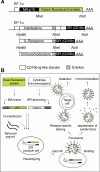
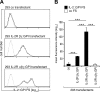
We describe for the first time fluorescent virus-like particles decorated with biologically active mono- and multisubunit immune receptors of choice and the basic application of such fluorosomes (FSs) to visualize and target immune receptor-ligand interactions. For that purpose, human embryonic kidney (HEK)-293 cells were stably transfected with Moloney murine leukemia virus (MoMLV) matrix protein (MA) GFP fusion constructs. To produce FSs, interleukins (ILs), IL-receptors (IL-Rs), and costimulatory molecules were fused to the glycosyl phosphatidyl inositol anchor acceptor sequence of CD16b and coexpressed along with MoMLV group-specific antigen-polymerase (gag-pol) in MA::GFP(+) HEK-293 cells. We show that IL-2 decorated but not control-decorated FSs specifically identify normal and malignant IL-2 receptor-positive (IL-2R(+)) lymphocytes by flow cytometry. In addition to cytokines and costimulatory molecules, FSs were also successfully decorated with the heterotrimeric IL-2Rs, allowing identification of IL-2(+) target cells. Specificity of binding was proven by complete inhibition with nonlabeled, soluble ligands. Moreover, IL-2R FSs efficiently neutralized soluble IL-2 and thus induced unresponsiveness of T cells receiving full activation stimuli via T-cell antigen receptor and CD28. FSs are technically simple, multivalent tools for assessing and blocking mono- and multisubunit immune receptor-ligand interactions with natural constituents in a plasma membrane context.
Figures



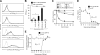
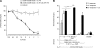
Similar articles
-
CD8+ T Cell Fate and Function Influenced by Antigen-Specific Virus-Like Nanoparticles Co-Expressing Membrane Tethered IL-2.PLoS One. 2015 May 6;10(5):e0126034. doi: 10.1371/journal.pone.0126034. eCollection 2015. PLoS One. 2015. PMID: 25946103 Free PMC article.
-
Fluorosomes: fluorescent virus-like nanoparticles that represent a convenient tool to visualize receptor-ligand interactions.Sensors (Basel). 2013 Jul 8;13(7):8722-49. doi: 10.3390/s130708722. Sensors (Basel). 2013. PMID: 23881135 Free PMC article. Review.
-
General strategy for decoration of enveloped viruses with functionally active lipid-modified cytokines.J Virol. 2007 Aug;81(16):8666-76. doi: 10.1128/JVI.00682-07. Epub 2007 May 30. J Virol. 2007. PMID: 17537846 Free PMC article.
-
Down-regulation of KOLT-2 antigen (CD28) by interleukin 2; role of IL-2R (p70).Immunology. 1990 Sep;71(1):63-9. Immunology. 1990. PMID: 2170270 Free PMC article.
-
Structure and function of IL-2 and IL-2 receptors.Behring Inst Mitt. 1992 Apr;(91):87-95. Behring Inst Mitt. 1992. PMID: 1524574 Review.
Cited by
-
Biomedical applications of glycosylphosphatidylinositol-anchored proteins.J Lipid Res. 2016 Oct;57(10):1778-1788. doi: 10.1194/jlr.R070201. Epub 2016 Aug 19. J Lipid Res. 2016. PMID: 27542385 Free PMC article. Review.
-
Past, present, and future of allergen immunotherapy vaccines.Allergy. 2021 Jan;76(1):131-149. doi: 10.1111/all.14300. Epub 2020 Apr 29. Allergy. 2021. PMID: 32249442 Free PMC article. Review.
-
The T-Cell Growth Factor Interleukin-2, Which Is Occasionally Targeted by Autoantibodies, Qualifies as Drug for the Treatment of Allergy, Autoimmunity, and Cancer: Collegium Internationale Allergologicum (CIA) Update 2024.Int Arch Allergy Immunol. 2024;185(3):286-300. doi: 10.1159/000533677. Epub 2023 Dec 12. Int Arch Allergy Immunol. 2024. PMID: 38086339 Free PMC article. Review.
-
CD8+ T Cell Fate and Function Influenced by Antigen-Specific Virus-Like Nanoparticles Co-Expressing Membrane Tethered IL-2.PLoS One. 2015 May 6;10(5):e0126034. doi: 10.1371/journal.pone.0126034. eCollection 2015. PLoS One. 2015. PMID: 25946103 Free PMC article.
-
Fluorosomes: fluorescent virus-like nanoparticles that represent a convenient tool to visualize receptor-ligand interactions.Sensors (Basel). 2013 Jul 8;13(7):8722-49. doi: 10.3390/s130708722. Sensors (Basel). 2013. PMID: 23881135 Free PMC article. Review.
References
-
- Derdak S V, Kueng H J, Leb V M, Neunkirchner A, Schmetterer K G, Bielek E, Majdic O, Knapp W, Seed B, Pickl W F. Direct stimulation of T lymphocytes by immunosomes: virus-like particles decorated with T cell receptor/CD3 ligands plus costimulatory molecules. Proc Natl Acad Sci U S A. 2006;103:13144–13149. - PMC - PubMed
-
- Leb V M, Jahn-Schmid B, Kueng H J, Schmetterer K G, Haiderer D, Neunkirchner A, Fischer G F, Hartl A, Thalhamer J, Steinberger P, Bohle B, Seed B, Pickl W F. Modulation of allergen-specific T-lymphocyte function by virus-like particles decorated with HLA class II molecules. J Allergy Clin Immunol. 2009;124:121–128. - PubMed
-
- Shimomura O, Johnson F H, Saiga Y. Extraction, purification and properties of aequorin, a bioluminescent protein from the luminous hydromedusan, Aequorea. J Cell Comp Physiol. 1962;59:223–239. - PubMed
-
- Haas J, Park E C, Seed B. Codon usage limitation in the expression of HIV-1 envelope glycoprotein. Curr Biol. 1996;6:315–324. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

