The microRNA-processing enzyme dicer maintains juxtaglomerular cells
- PMID: 20056748
- PMCID: PMC2831866
- DOI: 10.1681/ASN.2009090964
The microRNA-processing enzyme dicer maintains juxtaglomerular cells
Abstract
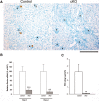
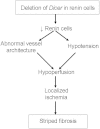
Juxtaglomerular cells are highly specialized myoepithelioid granulated cells located in the glomerular afferent arterioles. These cells synthesize and release renin, which distinguishes them from other cells. How these cells maintain their identity, restricted localization, and fate is unknown and is fundamental to the control of BP and homeostasis of fluid and electrolytes. Because microRNAs may control cell fate via temporal and spatial gene regulation, we generated mice with a conditional deletion of Dicer, the RNase III endonuclease that produces mature microRNAs in cells of the renin lineage. Deletion of Dicer severely reduced the number of juxtaglomerular cells, decreased expression of the renin genes (Ren1 and Ren2), lowered plasma renin concentration, and decreased BP. As a consequence of the disappearance of renin-producing cells, the kidneys developed striking vascular abnormalities and prominent striped fibrosis. We conclude that microRNAs maintain the renin-producing juxtaglomerular cells and the morphologic integrity and function of the kidney.
Figures




References
-
- Sequeira Lopez ML, Pentz ES, Nomasa T, Smithies O, Gomez RA: Renin cells are precursors for multiple cell types that switch to the renin phenotype when homeostasis is threatened. Dev Cell 6: 719–728, 2004 - PubMed
-
- Song L, Tuan RS: MicroRNAs and cell differentiation in mammalian development. Birth Defects Res C Embryo Today 78: 140–149, 2006 - PubMed
-
- Bartel DP: MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell 116: 281–297, 2004 - PubMed
-
- Stefani G, Slack FJ: Small non-coding RNAs in animal development. Nat Rev Mol Cell Biol 9: 219–230, 2008 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

