Diet-induced obesity in two C57BL/6 substrains with intact or mutant nicotinamide nucleotide transhydrogenase (Nnt) gene
- PMID: 20057372
- PMCID: PMC2888716
- DOI: 10.1038/oby.2009.477
Diet-induced obesity in two C57BL/6 substrains with intact or mutant nicotinamide nucleotide transhydrogenase (Nnt) gene
Abstract
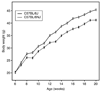
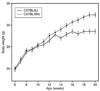
The C57BL/6J (B6/J) male mouse represents a standard for diet-induced obesity (DIO) and is unique in expressing a loss-of-function nicotinamide nucleotide transhydrogenase (Nnt) gene. This mutation was associated with a marked reduction in glucose-stimulated insulin secretion from B6/J islets in vitro and moderately impaired glucose clearance in vivo. To assess the contribution of this Nnt mutation, we compared DIO responsiveness of Nnt-mutant B6/J males to Nnt wild-type C57BL/6NJ (B6/NJ) males over a 14-week period of feeding a high-fat (60% of calories) diet. Initial mean body weights at 6 weeks did not distinguish the substrains and both substrains were DIO-sensitive. However, B6/J males outgained the B6/NJ males, with a significant 3 g higher mean body weight at 20 weeks accompanied by significant increases in both lean and fat mass. Mean nonfasting serum glucose over time was also significantly higher in B6/J males, as was impairment of glucose tolerance assessed at 8 and 20 weeks of age. Serum leptin, but not insulin, was significantly higher in B6/J males over time. Potential contributions of the wild-type Nnt gene were demonstrable on a lower fat diet (10% of calories) where a significantly greater weight gain over time by B6/NJ males was correlated with a significantly higher serum insulin. In conclusion, DIO developed in response to 60% fat feeding regardless of Nnt allele status. Contribution of the B6/J-unique Nnt mutation was most evident in response to 10% fat feeding that resulted in reduced serum insulin and weight gain compared to B6/NJ males.
Keywords: Adiposity; Animal Models; Dietary Fat; Genetics; Insulin Secretion.
Figures

Similar articles
-
The deletion variant of nicotinamide nucleotide transhydrogenase (Nnt) does not affect insulin secretion or glucose tolerance.Endocrinology. 2010 Jan;151(1):96-102. doi: 10.1210/en.2009-0887. Epub 2009 Nov 11. Endocrinology. 2010. PMID: 19906813
-
The lack of functional nicotinamide nucleotide transhydrogenase only moderately contributes to the impairment of glucose tolerance and glucose-stimulated insulin secretion in C57BL/6J vs C57BL/6N mice.Diabetologia. 2021 Nov;64(11):2550-2561. doi: 10.1007/s00125-021-05548-7. Epub 2021 Aug 27. Diabetologia. 2021. PMID: 34448880
-
Nicotinamide Nucleotide Transhydrogenase (Nnt) is Related to Obesity in Mice.Horm Metab Res. 2020 Dec;52(12):877-881. doi: 10.1055/a-1199-2257. Epub 2020 Jul 6. Horm Metab Res. 2020. PMID: 32629517
-
Deletion of nicotinamide nucleotide transhydrogenase: a new quantitive trait locus accounting for glucose intolerance in C57BL/6J mice.Diabetes. 2006 Jul;55(7):2153-6. doi: 10.2337/db06-0358. Diabetes. 2006. PMID: 16804088
-
Mitochondrial NAD(P)+ Transhydrogenase: From Molecular Features to Physiology and Disease.Antioxid Redox Signal. 2022 May;36(13-15):864-884. doi: 10.1089/ars.2021.0111. Epub 2021 Aug 5. Antioxid Redox Signal. 2022. PMID: 34155914 Review.
Cited by
-
Saturated Fatty Acid Activates T Cell Inflammation Through a Nicotinamide Nucleotide Transhydrogenase (NNT)-Dependent Mechanism.Biomolecules. 2019 Feb 25;9(2):79. doi: 10.3390/biom9020079. Biomolecules. 2019. PMID: 30823587 Free PMC article.
-
Impact of a novel homozygous mutation in nicotinamide nucleotide transhydrogenase on mitochondrial DNA integrity in a case of familial glucocorticoid deficiency.BBA Clin. 2015 Jun 1;3:70-78. doi: 10.1016/j.bbacli.2014.12.003. BBA Clin. 2015. PMID: 26309815 Free PMC article.
-
The interplay of maternal and offspring obesogenic diets: the impact on offspring metabolism and muscle mitochondria in an outbred mouse model.Front Physiol. 2024 Mar 22;15:1354327. doi: 10.3389/fphys.2024.1354327. eCollection 2024. Front Physiol. 2024. PMID: 38585221 Free PMC article.
-
Mouse Models of Gestational Diabetes Mellitus and Its Subtypes: Recent Insights and Pitfalls.Int J Mol Sci. 2023 Mar 22;24(6):5982. doi: 10.3390/ijms24065982. Int J Mol Sci. 2023. PMID: 36983056 Free PMC article. Review.
-
Nicotinamide riboside has minimal impact on energy metabolism in mouse models of mild obesity.J Endocrinol. 2021 Sep 9;251(1):111-123. doi: 10.1530/JOE-21-0123. J Endocrinol. 2021. PMID: 34370682 Free PMC article.
References
-
- Buettner R, Scholmerich J, Bollheimer LC. High-fat diets: modeling the metabolic disorders of human obesity in rodents. Obesity. 2007;15:798–808. - PubMed
-
- Collins S, Martin TL, Surwit RS, Robidoux J. Genetic vulnerability to dietinduced obesity in the C57BL/6J mouse: physiological and molecular characteristics. Physiol Behav. 2004;81:243–248. - PubMed
-
- Kaku K, Fiedorek F, Province M, Permutt M. Genetic analysis of glucose tolerance in inbred mouse strains. Diabetes. 1988;37:707–713. - PubMed
-
- Clee SM, Attie AD. The genetic landscape of type 2 diabetes in mice. Endocr Rev. 2007;28:48–83. - PubMed
-
- Toye AA, Lippiat JD, Proks P, et al. A genetic and physiological study of impaired glucose homeostasis control in C57BL/6J mice. Diabetologia. 2005;48:675–686. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

