Effects of acrylamide on the activity and structure of human brain creatine kinase
- PMID: 20057941
- PMCID: PMC2790104
- DOI: 10.3390/ijms10104210
Effects of acrylamide on the activity and structure of human brain creatine kinase
Abstract
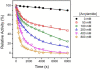
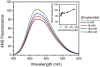
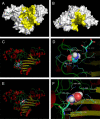
Acrylamide is widely used worldwide in industry and it can also be produced by the cooking and processing of foods. It is harmful to human beings, and human brain CK (HBCK) has been proposed to be one of the important targets of acrylamide. In this research, we studied the effects of acrylamide on HBCK activity, structure and the potential binding sites. Compared to CKs from rabbit, HBCK was fully inactivated at several-fold lower concentrations of acrylamide, and exhibited distinct properties upon acrylamide-induced inactivation and structural changes. The binding sites of acrylamide were located at the cleft between the N- and C-terminal domains of CK, and Glu232 was one of the key binding residues. The effects of acrylamide on CK were proposed to be isoenzyme- and species-specific, and the underlying molecular mechanisms were discussed.
Keywords: acrylamide; docking simulation; human brain creatine kinase; inactivation.
Figures


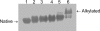
References
-
- Friedman M. Chemistry, biochemistry, and safety of acrylamide. A review. J. Agric. Food Chem. 2003;51:4504–4526. - PubMed
-
- Rydberg P, Eriksson S, Tareke E, Karlsson P, Ehrenberg L, Tornqvist M. Factors that influence the acrylamide content of heated foods. Adv. Exp. Med. Biol. 2005;561:317–328. - PubMed
-
- Tornqvist M. Acrylamide in food: The discovery and its implications: A historical perspective. Adv. Exp. Med. Biol. 2005;561:1–19. - PubMed
-
- Blank I. Current status of acrylamide research in food: Measurement, safety assessment, and formation. Ann. N. Y. Acad. Sci. 2005;1043:30–40. - PubMed
-
- Smith EA, Oehme FW. Acrylamide and polyacrylamide: A review of production, use, environmental fate and neurotoxicity. Rev. Environ. Health. 1991;9:215–228. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

