Hyperoxic reperfusion after global cerebral ischemia promotes inflammation and long-term hippocampal neuronal death
- PMID: 20059303
- PMCID: PMC2867550
- DOI: 10.1089/neu.2009.1186
Hyperoxic reperfusion after global cerebral ischemia promotes inflammation and long-term hippocampal neuronal death
Abstract
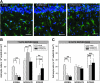
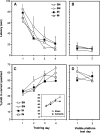
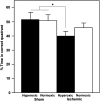
In this study we tested the hypothesis that long-term neuropathological outcome is worsened by hyperoxic compared to normoxic reperfusion in a rat global cerebral ischemia model. Adult male rats were anesthetized and subjected to bilateral carotid arterial occlusion plus bleeding hypotension for 10 min. The rats were randomized to one of four protocols: ischemia/normoxia (21% oxygen for 1 h), ischemia/hyperoxia (100% oxygen for 1 h), sham/normoxia, and sham/hyperoxia. Hippocampal CA1 neuronal survival and activation of microglia and astrocytes were measured in the hippocampi of the animals at 7 and 30 days post-ischemia. Morris water maze testing of memory was performed on days 23-30. Compared to normoxic reperfusion, hyperoxic ventilation resulted in a significant decrease in normal-appearing neurons at 7 and 30 days, and increased activation of microglia and astrocytes at 7, but not at 30, days of reperfusion. Behavioral deficits were also observed following hyperoxic, but not normoxic, reperfusion. We conclude that early post-ischemic hyperoxic reperfusion is followed by greater hippocampal neuronal death and cellular inflammatory reactions compared to normoxic reperfusion. The results of these long-term outcome studies, taken together with previously published results from short-term experiments performed with large animals, support the hypothesis that neurological outcome can be improved by avoiding hyperoxic resuscitation after global cerebral ischemia such as that which accompanies cardiac arrest.
Figures


References
-
- Dietrich W.D. Busto R. Alonso O. Globus M.Y. Ginsberg M.D. Intraischemic but not postischemic brain hypothermia protects chronically following global forebrain ischemia in rats. J. Cereb. Blood Flow Metab. 1993;13:541–549. - PubMed
-
- Feng Z.C. Sick T.J. Rosenthal M. Oxygen sensitivity of mitochondrial redox status and evoked potential recovery early during reperfusion in post-ischemic rat brain. Resuscitation. 1998;37:33–41. - PubMed
-
- Hailer N.P. Immunosuppression after traumatic or ischemic CNS damage: it is neuroprotective and illuminates the role of microglial cells. Prog. Neurobiol. 2008;84:211–233. - PubMed
-
- Liebeskind D.S. Kasner S.E. Neuroprotection for ischaemic stroke: an unattainable goal? CNS Drugs. 2001;15:165–174. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

