ICC-MY coordinate smooth muscle electrical and mechanical activity in the murine small intestine
- PMID: 20059699
- PMCID: PMC2856807
- DOI: 10.1111/j.1365-2982.2009.01448.x
ICC-MY coordinate smooth muscle electrical and mechanical activity in the murine small intestine
Abstract
Background: Animals carrying genetic mutations have provided powerful insights into the role of interstitial cells of Cajal (ICC) in motility. One classic model is the W/W(V) mouse which carries loss-of-function mutations in c-kit alleles, but retains minimal function of the tyrosine kinase. Previous studies have documented loss of slow waves and aberrant motility in the small intestine of W/W(V) mice where myenteric ICC (ICC-MY) are significantly depleted.
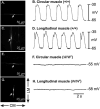
Methods: Here, we used morphological and electrophysiological techniques to further assess the loss of ICC around the circumference of the small intestine and determine consequences of losing ICC-MY on electrical activity, Ca(2+) transients and contractions of the longitudinal muscle (LM).
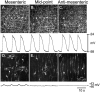
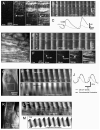
Key results: In wild-type mice, there was coherent propagation of Ca(2+) transients through the ICC-MY network and spread of this activity to the LM. In short segments of small intestine in vitro and in exteriorized segments, slow waves coordinated smoothly propagating Ca(2+) waves and contractions in the LM of wild-type mice. In W/W(V) mice, Ca(2+) waves were initiated at variable sites along and around intestinal segments and propagated without constraint unless they collided with other Ca(2+) waves. This activity resulted in abrupt, uncoordinated contractions.
Conclusions & inferences: These results show how dominance of pacemaking by ICC-MY coordinates propagating con-tractions and regulates the spontaneous activity of smooth muscle.
Conflict of interest statement
Competing Interests: the authors have no competing interests.
Figures




References
-
- Vanderwinden JM, Rumessen JJ. Interstitial cells of Cajal in human gut and gastrointestinal disease. Microsc Res Tech. 1999;47:344–360. - PubMed
-
- Sanders KM, Ordög T, Koh SD, Torihashi S, Ward SM. Development and plasticity of interstitial cells of Cajal. Neurogastroenterol Motil. 1999;11:311–38. - PubMed
-
- Burns AJ. Disorders of interstitial cells of Cajal. J Pediatr Gastroenterol Nutr. 2007;45:103–6. - PubMed
-
- Rolle U, Piaseczna-Piotrowska A, Puri P. Interstitial cells of Cajal in the normal gut and in intestinal motility disorders of childhood. Pediatr Surg Int. 2007;23:1139–52. - PubMed
-
- Farrugia G. Interstitial cells of Cajal in health and disease. Neurogastroenterol Motil. 2008;20:54–63. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

