Delayed enrichment of mesenchymal cells promotes cardiac lineage and calcium transient development
- PMID: 20060001
- PMCID: PMC2837799
- DOI: 10.1016/j.yjmcc.2009.12.022
Delayed enrichment of mesenchymal cells promotes cardiac lineage and calcium transient development
Abstract
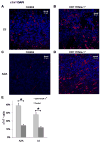
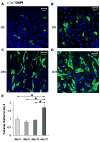
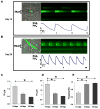
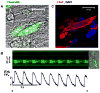
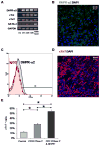
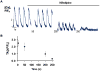
Bone marrow-derived mesenchymal stem cells (BM-MSCs) can be induced to differentiate into myogenic cells. Despite their potential, previous studies have not been successful in producing a high percentage of cardiac-like cells with a muscle phenotype. We hypothesized that cardiac lineage development in BM-MSC is related to cell passage, culture milieu, and enrichment for specific cell subtypes before and during differentiation. Our study demonstrated that Lin(-) BM-MSC at an intermediate passage (IP; P8-P12) expressed cardiac troponin T (cTnT) after 21 days in culture. Cardiac TnT expression was similar whether IP cells were differentiated in media containing 5-azacytidine+2% FBS (AZA; 14%) or 2% FBS alone (LS; 12%) and both were significantly higher than AZA+5% FBS. This expression was potentiated by first enriching for CD117/Sca-1 cells followed by differentiation (AZA, 39% and LS, 28%). A second sequential enrichment for the dihydropyridine receptor subunit alpha2delta1 (DHPR-alpha2) resulted in cardiac TnT expressed in 54% of cultured cells compared to 28% of cells after CD117/Sca-1(+) enrichment. Cells enriched for CD117/Sca-1 and subjected to differentiation displayed spontaneous intracellular Ca(2+) transients with an increase in transient frequency and a 60% decrease in the transient duration amplitude between days 14 and 29. In conclusion, IP CD117/Sca-1(+) murine BM-MSCs display robust cardiac muscle lineage development that can be induced independent of AZA but is diminished under higher serum concentrations. Furthermore, temporal changes in calcium kinetics commensurate with increased cTnT expression suggest progressive maturation of a cardiac muscle lineage. Enrichment with CD117/Sca-1 to establish lineage commitment followed by DHPR-alpha2 in lineage developing cells may enhance the therapeutic potential of these cells for transplantation.
Copyright (c) 2009 Elsevier Ltd. All rights reserved.
Figures

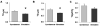
Similar articles
-
Evaluation of the effects of different culture media on the myogenic differentiation potential of adipose tissue- or bone marrow-derived human mesenchymal stem cells.Int J Mol Med. 2014 Jan;33(1):160-70. doi: 10.3892/ijmm.2013.1555. Epub 2013 Nov 13. Int J Mol Med. 2014. PMID: 24220225
-
Differentiation of mesenchymal stem cells derived from pancreatic islets and bone marrow into islet-like cell phenotype.PLoS One. 2011;6(12):e28175. doi: 10.1371/journal.pone.0028175. Epub 2011 Dec 16. PLoS One. 2011. PMID: 22194812 Free PMC article.
-
Delayed enrichment for c-kit and inducing cardiac differentiation attenuated protective effects of BMSCs' transplantation in pig model of acute myocardial ischemia.Cardiovasc Ther. 2015 Aug;33(4):184-92. doi: 10.1111/1755-5922.12131. Cardiovasc Ther. 2015. PMID: 25959676
-
Mesenchymal stem cells from adult human bone marrow differentiate into a cardiomyocyte phenotype in vitro.Exp Biol Med (Maywood). 2004 Jul;229(7):623-31. doi: 10.1177/153537020422900706. Exp Biol Med (Maywood). 2004. PMID: 15229356
-
Calcium-dependent potassium channels control proliferation of cardiac progenitor cells and bone marrow-derived mesenchymal stem cells.J Physiol. 2018 Jun;596(12):2359-2379. doi: 10.1113/JP275388. Epub 2018 May 5. J Physiol. 2018. PMID: 29574723 Free PMC article.
Cited by
-
In vitro differentiation of W8B2+ human cardiac stem cells: gene expression of ionic channels and spontaneous calcium activity.Cell Mol Biol Lett. 2020 Nov 5;25(1):50. doi: 10.1186/s11658-020-00242-9. Cell Mol Biol Lett. 2020. PMID: 33292162 Free PMC article.
-
Ischemia/Reperfusion injury protection by mesenchymal stem cell derived antioxidant capacity.Stem Cells Dev. 2013 Sep 15;22(18):2497-507. doi: 10.1089/scd.2013.0136. Epub 2013 Jun 11. Stem Cells Dev. 2013. PMID: 23614555 Free PMC article.
-
Effect of serum and oxygen concentration on gene expression and secretion of paracrine factors by mesenchymal stem cells.Int J Cell Biol. 2014;2014:601063. doi: 10.1155/2014/601063. Epub 2014 Dec 29. Int J Cell Biol. 2014. PMID: 25614742 Free PMC article.
-
Ca2+ dysregulation in cardiac stromal cells sustains fibro-adipose remodeling in Arrhythmogenic Cardiomyopathy and can be modulated by flecainide.J Transl Med. 2022 Nov 12;20(1):522. doi: 10.1186/s12967-022-03742-8. J Transl Med. 2022. PMID: 36371290 Free PMC article.
-
Mesenchymal stem cells improve cardiac conduction by upregulation of connexin 43 through paracrine signaling.Am J Physiol Heart Circ Physiol. 2013 Feb 15;304(4):H600-9. doi: 10.1152/ajpheart.00533.2012. Epub 2012 Dec 15. Am J Physiol Heart Circ Physiol. 2013. PMID: 23241322 Free PMC article.
References
-
- Cimini M, Fazel S, Zhuo S, Xaymardan M, Fujii H, Weisel RD, et al. c-kit dysfunction impairs myocardial healing after infarction. Circulation. 2007;116(11 Suppl):I77–82. - PubMed
-
- Lutz M, Rosenberg M, Kiessling F, Eckstein V, Heger T, Krebs J, et al. Local injection of stem cell factor (SCF) improves myocardial homing of systemically delivered c-kit + bone marrow-derived stem cells. Cardiovasc Res. 2008;77(1):143–50. - PubMed
-
- Okada S, Nakauchi H, Nagayoshi K, Nishikawa S, Miura Y, Suda T. In vivo and in vitro stem cell function of c-kit- and Sca-1-positive murine hematopoietic cells. Blood. 1992;80(12):3044–50. - PubMed
-
- Thoren LA, Liuba K, Bryder D, Nygren JM, Jensen CT, Qian H, et al. Kit regulates maintenance of quiescent hematopoietic stem cells. J Immunol. 2008;180(4):2045–53. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

