Fenofibrate reduces systemic inflammation markers independent of its effects on lipid and glucose metabolism in patients with the metabolic syndrome
- PMID: 20061429
- PMCID: PMC2840858
- DOI: 10.1210/jc.2009-1487
Fenofibrate reduces systemic inflammation markers independent of its effects on lipid and glucose metabolism in patients with the metabolic syndrome
Abstract
Context: Fenofibrate is a peroxisome proliferator-activated receptor alpha agonist widely used in clinical practice, but its mechanism of action is incompletely understood.
Objective: The aim of the study was to assess whether improvement in subclinical inflammation or glucose metabolism contributes to its antiatherogenic effects in insulin-resistant subjects with the metabolic syndrome (MetS).
Design and setting: We conducted a randomized, double-blind, placebo-controlled study in the research unit at an academic center.
Patients: We studied 25 nondiabetic insulin-resistant MetS subjects.
Intervention(s): We administered fenofibrate (200 mg/d) and placebo for 12 wk.
Main outcome measures: Before and after treatment, we measured plasma lipids/apolipoproteins, inflammatory markers (high-sensitivity C-reactive protein, IL-6, intercellular adhesion molecule/vascular cell adhesion molecule), adipocytokines (adiponectin, TNFalpha, leptin), and insulin secretion (oral glucose tolerance test). We also assessed adipose tissue, hepatic and peripheral (muscle) insulin resistance fasting and during a euglycemic insulin clamp with (3)H glucose and (14)C palmitate infusion combined with indirect calorimetry.
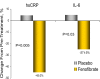
Results: Subjects displayed severe insulin resistance and systemic inflammation. Fenofibrate significantly reduced plasma triglyceride, apolipoprotein (apo) CII, apo CIII, and apo E (all P < 0.01), with a modest increase in high-density lipoprotein-cholesterol (+12%; P = 0.06). Fenofibrate markedly decreased plasma high-sensitivity C-reactive protein by 49.5 +/- 8% (P = 0.005) and IL-6 by 29.8 +/- 7% (P = 0.03) vs. placebo. However, neither insulin secretion nor adipose tissue, hepatic or muscle insulin sensitivity or glucose/lipid oxidation improved with treatment. Adiponectin and TNF-alpha levels were also unchanged. Improvement in plasma markers of vascular/systemic inflammation was dissociated from changes in triglyceride/high-density lipoprotein-cholesterol, apo CII/CIII, or free fatty acid concentrations or insulin secretion/insulin sensitivity.
Conclusions: In subjects with the MetS, fenofibrate reduces systemic inflammation independent of improvements in lipoprotein metabolism and without changing insulin sensitivity. This suggests a direct peroxisome proliferator-activated receptor alpha-mediated effect of fenofibrate on inflammatory pathways, which may be important for the prevention of CVD in high-risk patients.
Figures
Similar articles
-
Free fatty acid metabolism during fenofibrate treatment of the metabolic syndrome.Clin Pharmacol Ther. 2003 Sep;74(3):236-44. doi: 10.1016/S0009-9236(03)00170-X. Clin Pharmacol Ther. 2003. PMID: 12966367 Clinical Trial.
-
Effect of fenofibrate and niacin on intrahepatic triglyceride content, very low-density lipoprotein kinetics, and insulin action in obese subjects with nonalcoholic fatty liver disease.J Clin Endocrinol Metab. 2010 Jun;95(6):2727-35. doi: 10.1210/jc.2009-2622. Epub 2010 Apr 6. J Clin Endocrinol Metab. 2010. PMID: 20371660 Free PMC article. Clinical Trial.
-
Peroxisome proliferator-activated receptor-alpha selective ligand reduces adiposity, improves insulin sensitivity and inhibits atherosclerosis in LDL receptor-deficient mice.Mol Cell Biochem. 2006 Apr;285(1-2):35-50. doi: 10.1007/s11010-005-9053-y. Epub 2006 Feb 14. Mol Cell Biochem. 2006. PMID: 16477380
-
Role of the PPAR-α agonist fenofibrate in severe pediatric burn.Burns. 2012 Jun;38(4):481-6. doi: 10.1016/j.burns.2011.12.004. Epub 2012 Jan 9. Burns. 2012. PMID: 22226866 Free PMC article. Review.
-
Fenofibrate: a review of its use in primary dyslipidaemia, the metabolic syndrome and type 2 diabetes mellitus.Drugs. 2007;67(1):121-53. doi: 10.2165/00003495-200767010-00013. Drugs. 2007. PMID: 17209672 Review.
Cited by
-
HIV protease inhibitors disrupt lipid metabolism by activating endoplasmic reticulum stress and inhibiting autophagy activity in adipocytes.PLoS One. 2013;8(3):e59514. doi: 10.1371/journal.pone.0059514. Epub 2013 Mar 22. PLoS One. 2013. PMID: 23533630 Free PMC article.
-
Effects of pemafibrate on glucose metabolism markers and liver function tests in patients with hypertriglyceridemia: a pooled analysis of six phase 2 and phase 3 randomized double-blind placebo-controlled clinical trials.Cardiovasc Diabetol. 2021 May 4;20(1):96. doi: 10.1186/s12933-021-01291-w. Cardiovasc Diabetol. 2021. PMID: 33947390 Free PMC article.
-
Dual peroxisome proliferator-activated receptor α/δ agonist GFT505 improves hepatic and peripheral insulin sensitivity in abdominally obese subjects.Diabetes Care. 2013 Oct;36(10):2923-30. doi: 10.2337/dc12-2012. Epub 2013 May 28. Diabetes Care. 2013. PMID: 23715754 Free PMC article. Clinical Trial.
-
Atheroprotective effect of oleoylethanolamide (OEA) targeting oxidized LDL.PLoS One. 2014 Jan 20;9(1):e85337. doi: 10.1371/journal.pone.0085337. eCollection 2014. PLoS One. 2014. PMID: 24465540 Free PMC article.
-
Clinical approach to the inflammatory etiology of cardiovascular diseases.Pharmacol Res. 2020 Sep;159:104916. doi: 10.1016/j.phrs.2020.104916. Epub 2020 May 20. Pharmacol Res. 2020. PMID: 32445957 Free PMC article. Review.
References
-
- Cusi K 2001 Cardiovascular risk management in type 2 diabetes: from clinical trials to clinical practice. The Endocrinologist 11:474–490
-
- Caballero AE 2004 Endothelial dysfunction, inflammation, and insulin resistance: a focus on subjects at risk for type 2 diabetes. Curr Diab Rep 4:237–246 - PubMed
-
- Frick MH, Elo O, Haapa K, Heinonen OP, Heinsalmi P, Helo P, Huttunen JK, Kaitaniemi P, Koskinen P, Manninen V. Helsinki Heart Study 1987 Primary-prevention trial with gemfibrozil in middle-aged men with dyslipidemia. Safety of treatment, changes in risk factors, and incidence of coronary heart disease. N Engl J Med 317:1237–1245 - PubMed
-
- Keech A, Simes RJ, Barter P, Best J, Scott R, Taskinen MR, Forder P, Pillai A, Davis T, Glasziou P, Drury P, Kesäniemi YA, Sullivan D, Hunt D, Colman P, d'Emden M, Whiting M, Ehnholm C, Laakso M 2005 Effects of long-term fenofibrate therapy on cardiovascular events in 9795 people with type 2 diabetes mellitus (the FIELD study): randomised controlled trial. Lancet 366:1849–1861 - PubMed
-
- Rubins HB, Robins SJ, Collins D, Nelson DB, Elam MB, Schaefer EJ, Faas FH, Anderson JW for the VA-HIT Study Group 2002 Diabetes, plasma insulin and cardiovascular disease: subgroup analysis from the Department of Veterans Affairs High-Density Lipoprotein Intervention Trial (VA-HIT). Arch Intern Med 162:2597–2604 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials