Glycosylation of the collagen adhesin EmaA of Aggregatibacter actinomycetemcomitans is dependent upon the lipopolysaccharide biosynthetic pathway
- PMID: 20061477
- PMCID: PMC2820841
- DOI: 10.1128/JB.01453-09
Glycosylation of the collagen adhesin EmaA of Aggregatibacter actinomycetemcomitans is dependent upon the lipopolysaccharide biosynthetic pathway
Abstract
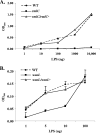
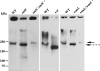
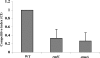
The human oropharyngeal pathogen Aggregatibacter actinomycetemcomitans synthesizes multiple adhesins, including the nonfimbrial extracellular matrix protein adhesin A (EmaA). EmaA monomers trimerize to form antennae-like structures on the surface of the bacterium, which are required for collagen binding. Two forms of the protein have been identified, which are suggested to be linked with the type of O-polysaccharide (O-PS) of the lipopolysaccharide (LPS) synthesized (G. Tang et al., Microbiology 153:2447-2457, 2007). This association was investigated by generating individual mutants for a rhamnose sugar biosynthetic enzyme (rmlC; TDP-4-keto-6-deoxy-d-glucose 3,5-epimerase), the ATP binding cassette (ABC) sugar transport protein (wzt), and the O-antigen ligase (waaL). All three mutants produced reduced amounts of O-PS, and the EmaA monomers in these mutants displayed a change in their electrophoretic mobility and aggregation state, as observed in sodium dodecyl sulfate (SDS)-polyacrylamide gels. The modification of EmaA with O-PS sugars was suggested by lectin blots, using the fucose-specific Lens culinaris agglutinin (LCA). Fucose is one of the glycan components of serotype b O-PS. The rmlC mutant strain expressing the modified EmaA protein demonstrated reduced collagen adhesion using an in vitro rabbit heart valve model, suggesting a role for the glycoconjugant in collagen binding. These data provide experimental evidence for the glycosylation of an oligomeric, coiled-coil adhesin and for the dependence of the posttranslational modification of EmaA on the LPS biosynthetic machinery in A. actinomycetemcomitans.
Figures
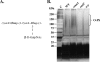



Similar articles
-
O-polysaccharide glycosylation is required for stability and function of the collagen adhesin EmaA of Aggregatibacter actinomycetemcomitans.Infect Immun. 2012 Aug;80(8):2868-77. doi: 10.1128/IAI.00372-12. Epub 2012 Jun 11. Infect Immun. 2012. PMID: 22689812 Free PMC article.
-
Dual function of the O-antigen WaaL ligase of Aggregatibacter actinomycetemcomitans.Mol Oral Microbiol. 2023 Dec;38(6):471-488. doi: 10.1111/omi.12444. Epub 2023 Nov 8. Mol Oral Microbiol. 2023. PMID: 37941494 Free PMC article.
-
Serotype-Specific Sugars Impact Structure but Not Functions of the Trimeric Autotransporter Adhesin EmaA of Aggregatibacter actinomycetemcomitans.J Bacteriol. 2022 Dec 20;204(12):e0021522. doi: 10.1128/jb.00215-22. Epub 2022 Nov 30. J Bacteriol. 2022. PMID: 36448790 Free PMC article.
-
The Trimeric Autotransporter Adhesin EmaA and Infective Endocarditis.Pathogens. 2024 Jan 23;13(2):99. doi: 10.3390/pathogens13020099. Pathogens. 2024. PMID: 38392837 Free PMC article. Review.
-
Synthesis of lipopolysaccharide O-antigens by ABC transporter-dependent pathways.Carbohydr Res. 2012 Jul 15;356:12-24. doi: 10.1016/j.carres.2012.02.027. Epub 2012 Mar 6. Carbohydr Res. 2012. PMID: 22475157 Review.
Cited by
-
The serotype a-EmaA adhesin of Aggregatibacter actinomycetemcomitans does not require O-PS synthesis for collagen binding activity.Microbiology (Reading). 2022 May;168(5):001191. doi: 10.1099/mic.0.001191. Microbiology (Reading). 2022. PMID: 35551696 Free PMC article.
-
Unconventional N-Linked Glycosylation Promotes Trimeric Autotransporter Function in Kingella kingae and Aggregatibacter aphrophilus.mBio. 2015 Aug 25;6(4):e01206-15. doi: 10.1128/mBio.01206-15. mBio. 2015. PMID: 26307167 Free PMC article.
-
O-polysaccharide glycosylation is required for stability and function of the collagen adhesin EmaA of Aggregatibacter actinomycetemcomitans.Infect Immun. 2012 Aug;80(8):2868-77. doi: 10.1128/IAI.00372-12. Epub 2012 Jun 11. Infect Immun. 2012. PMID: 22689812 Free PMC article.
-
Dual function of the O-antigen WaaL ligase of Aggregatibacter actinomycetemcomitans.Mol Oral Microbiol. 2023 Dec;38(6):471-488. doi: 10.1111/omi.12444. Epub 2023 Nov 8. Mol Oral Microbiol. 2023. PMID: 37941494 Free PMC article.
-
ABC transporters involved in export of cell surface glycoconjugates.Microbiol Mol Biol Rev. 2010 Sep;74(3):341-62. doi: 10.1128/MMBR.00009-10. Microbiol Mol Biol Rev. 2010. PMID: 20805402 Free PMC article. Review.
References
-
- Bengoechea, J., H. Najdenski, and M. Skurnik. 2004. Lipopolysaccharide O antigen status of Yersinia enterocolitica O:8 is essential for virulence and absence of O antigen affects the expression of other Yersinia virulence factors. Mol. Microbiol. 52:451-469. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

