Anti-tumor activity of stability-engineered IgG-like bispecific antibodies targeting TRAIL-R2 and LTbetaR
- PMID: 20061822
- PMCID: PMC2725416
- DOI: 10.4161/mabs.1.2.7631
Anti-tumor activity of stability-engineered IgG-like bispecific antibodies targeting TRAIL-R2 and LTbetaR
Abstract
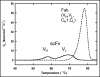
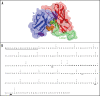
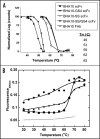
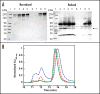
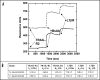
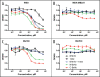
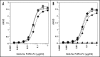
Bispecific antibodies (BsAbs) represent an emerging class of biologics that achieve dual targeting with a single agent. Recombinant DNA technologies have facilitated a variety of creative bispecific designs with many promising therapeutic applications; however, practical methods for producing high quality BsAbs that have good product stability, long serum half-life, straightforward purification, and scalable production have largely been limiting. Here we describe a protein-engineering approach for producing stable, scalable tetravalent IgG-like BsAbs. The stability-engineered IgG-like BsAb was envisioned to target and crosslink two TNF family member receptors, TRAIL-R2 (TNF-Related Apoptosis Inducing Ligand Receptor-2) and LTbetaR (Lymphotoxin-beta Receptor), expressed on the surface of epithelial tumor cells with the goal of triggering an enhanced anti-tumor effect. Our IgG-like BsAbs consists of a stability-engineered anti-LTbetaR single chain Fv (scFv) genetically fused to either the N- or C-terminus of the heavy chain of a fulllength anti-TRAIL-R2 IgG1 monoclonal antibody. Both N- or C-terminal BsAbs were active in inhibiting tumor cell growth in vitro, and with some cell lines demonstrated enhanced activity relative to the combination of parental Abs. Pharmacokinetic studies in mice revealed long serum half-lives for the BsAbs. In murine tumor xenograft models, therapeutic treatment with the BsAbs resulted in reduction in tumor volume either comparable to or greater than the combination of parental antibodies, indicating that simultaneously targeting and cross-linking receptor pairs is an effective strategy for treating tumor cells. These studies support that stability-engineering is an enabling step for producing scalable IgG-like BsAbs with properties desirable for biopharmaceutical development.
Figures


References
-
- Fischer N, Leger O. Bispecific antibodies: molecules that enable novel therapeutic strategies. Pathobiology. 2007;74:3–14. - PubMed
-
- Boerman OC, van Schaijk FG, Oyen WJ, Corstens FH. Pretargeted radioimmunotherapy of cancer: progress step by step. J Nucl Med. 2003;44:400–411. - PubMed
-
- Jain M, Kamal N, Batra SK. Engineering antibodies for clinical applications. Trends Biotechnol. 2007;25:307–316. - PubMed
-
- Kontermann RE. Recombinant bispecific antibodies for cancer therapy. Acta Pharmacol Sin. 2005;26:1–9. - PubMed
-
- Marvin JS, Zhu Z. Recombinant approaches to IgG-like bispecific antibodies. Acta Pharmacol Sin. 2005;26:649–658. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
