HCN channels are not required for mechanotransduction in sensory hair cells of the mouse inner ear
- PMID: 20062532
- PMCID: PMC2797612
- DOI: 10.1371/journal.pone.0008627
HCN channels are not required for mechanotransduction in sensory hair cells of the mouse inner ear
Abstract
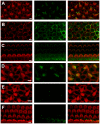
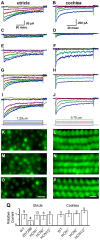
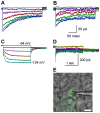
The molecular composition of the hair cell transduction channel has not been identified. Here we explore the novel hypothesis that hair cell transduction channels include HCN subunits. The HCN family of ion channels includes four members, HCN1-4. They were originally identified as the molecular correlates of the hyperpolarization-activated, cyclic nucleotide gated ion channels that carry currents known as If, IQ or Ih. However, based on recent evidence it has been suggested that HCN subunits may also be components of the elusive hair cell transduction channel. To investigate this hypothesis we examined expression of mRNA that encodes HCN1-4 in sensory epithelia of the mouse inner ear, immunolocalization of HCN subunits 1, 2 and 4, uptake of the transduction channel permeable dye, FM1-43 and electrophysiological measurement of mechanotransduction current. Dye uptake and transduction current were assayed in cochlear and vestibular hair cells of wildtype mice exposed to HCN channel blockers or a dominant-negative form of HCN2 that contained a pore mutation and in mutant mice that lacked HCN1, HCN2 or both. We found robust expression of HCNs 1, 2 and 4 but little evidence that localized HCN subunits in hair bundles, the site of mechanotransduction. Although high concentrations of the HCN antagonist, ZD7288, blocked 50-70% of the transduction current, we found no reduction of transduction current in either cochlear or vestibular hair cells of HCN1- or HCN2- deficient mice relative to wild-type mice. Furthermore, mice that lacked both HCN1 and HCN2 also had normal transduction currents. Lastly, we found that mice exposed to the dominant-negative mutant form of HCN2 had normal transduction currents as well. Taken together, the evidence suggests that HCN subunits are not required for mechanotransduction in hair cells of the mouse inner ear.
Conflict of interest statement
Figures

Similar articles
-
HCN channels expressed in the inner ear are necessary for normal balance function.J Neurosci. 2011 Nov 16;31(46):16814-25. doi: 10.1523/JNEUROSCI.3064-11.2011. J Neurosci. 2011. PMID: 22090507 Free PMC article.
-
Mechanotransduction and hyperpolarization-activated currents contribute to spontaneous activity in mouse vestibular ganglion neurons.J Gen Physiol. 2014 Apr;143(4):481-97. doi: 10.1085/jgp.201311126. Epub 2014 Mar 17. J Gen Physiol. 2014. PMID: 24638995 Free PMC article.
-
HCN1 and HCN2 proteins are expressed in cochlear hair cells: HCN1 can form a ternary complex with protocadherin 15 CD3 and F-actin-binding filamin A or can interact with HCN2.J Biol Chem. 2012 Nov 2;287(45):37628-46. doi: 10.1074/jbc.M112.375832. Epub 2012 Sep 4. J Biol Chem. 2012. PMID: 22948144 Free PMC article.
-
Transmembrane channel-like (TMC) genes are required for auditory and vestibular mechanosensation.Pflugers Arch. 2015 Jan;467(1):85-94. doi: 10.1007/s00424-014-1582-3. Epub 2014 Jul 31. Pflugers Arch. 2015. PMID: 25074487 Free PMC article. Review.
-
Pathophysiology of HCN channels.Pflugers Arch. 2007 Jul;454(4):517-22. doi: 10.1007/s00424-007-0224-4. Epub 2007 Feb 14. Pflugers Arch. 2007. PMID: 17549513 Review.
Cited by
-
Hyperpolarization-activated current (I(h)) in vestibular calyx terminals: characterization and role in shaping postsynaptic events.J Assoc Res Otolaryngol. 2012 Dec;13(6):745-58. doi: 10.1007/s10162-012-0342-3. Epub 2012 Jul 24. J Assoc Res Otolaryngol. 2012. PMID: 22825486 Free PMC article.
-
HCN channels in the mammalian cochlea: Expression pattern, subcellular location, and age-dependent changes.J Neurosci Res. 2021 Feb;99(2):699-728. doi: 10.1002/jnr.24754. Epub 2020 Nov 12. J Neurosci Res. 2021. PMID: 33181864 Free PMC article.
-
Uncovering genomic features and maternal origin of korean native chicken by whole genome sequencing.PLoS One. 2014 Dec 11;9(12):e114763. doi: 10.1371/journal.pone.0114763. eCollection 2014. PLoS One. 2014. PMID: 25501044 Free PMC article.
-
A comprehensive review of HCN channel expression and Ih in the auditory system: then, now, and future perspectives.J Neurophysiol. 2025 Aug 1;134(2):458-470. doi: 10.1152/jn.00602.2024. Epub 2025 Jul 7. J Neurophysiol. 2025. PMID: 40623052 Free PMC article. Review.
-
Neural substrates, dynamics and thresholds of galvanic vestibular stimulation in the behaving primate.Nat Commun. 2019 Apr 23;10(1):1904. doi: 10.1038/s41467-019-09738-1. Nat Commun. 2019. PMID: 31015434 Free PMC article.
References
-
- Kazmierczak P, Sakaguchi H, Tokita J, Wilson-Kubalek EM, Milligan RA, et al. Cadherin 23 and protocadherin 15 interact to form tip-link filaments in sensory hair cells. Nature. 2007;449:87–91. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

