Gene expression profile of androgen modulated genes in the murine fetal developing lung
- PMID: 20064212
- PMCID: PMC2822783
- DOI: 10.1186/1477-7827-8-2
Gene expression profile of androgen modulated genes in the murine fetal developing lung
Abstract
Background: Accumulating evidences suggest that sex affects lung development. Indeed, a higher incidence of respiratory distress syndrome is observed in male compared to female preterm neonates at comparable developmental stage and experimental studies demonstrated an androgen-related delay in male lung maturation. However, the precise mechanisms underlying these deleterious effects of androgens in lung maturation are only partially understood.
Methods: To build up a better understanding of the effect of androgens on lung development, we analyzed by microarrays the expression of genes showing a sexual difference and those modulated by androgens. Lungs of murine fetuses resulting from a timely mating window of 1 hour were studied at gestational day 17 (GD17) and GD18, corresponding to the period of surge of surfactant production. Using injections of the antiandrogen flutamide to pregnant mice, we hunted for genes in fetal lungs which are transcriptionally modulated by androgens.
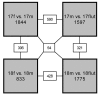
Results: Results revealed that 1844 genes were expressed with a sexual difference at GD17 and 833 at GD18. Many genes were significantly modulated by flutamide: 1597 at GD17 and 1775 at GD18. Datasets were analyzed by using in silico tools for reconstruction of cellular pathways. Between GD17 and GD18, male lungs showed an intensive transcriptional activity of proliferative pathways along with the onset of lung differentiation. Among the genes showing a sex difference or an antiandrogen modulation of their expression, we specifically identified androgen receptor interacting genes, surfactant related genes in particularly those involved in the pathway leading to phospholipid synthesis, and several genes of lung development regulator pathways. Among these latter, some genes related to Shh, FGF, TGF-beta, BMP, and Wnt signaling are modulated by sex and/or antiandrogen treatment.
Conclusion: Our results show clearly that there is a real delay in lung maturation between male and female in this period, the latter pursuing already lung maturation while the proper is not yet fully engaged in the differentiation processes at GD17. In addition, this study provides a list of genes which are under the control of androgens within the lung at the moment of surge of surfactant production in murine fetal lung.
Figures



Similar articles
-
Sexually dimorphic gene expression that overlaps maturation of type II pneumonocytes in fetal mouse lungs.Reprod Biol Endocrinol. 2006 May 4;4:25. doi: 10.1186/1477-7827-4-25. Reprod Biol Endocrinol. 2006. PMID: 16674826 Free PMC article.
-
Expression profile of androgen-modulated microRNAs in the fetal murine lung.Biol Sex Differ. 2016 Apr 1;7:20. doi: 10.1186/s13293-016-0072-z. eCollection 2016. Biol Sex Differ. 2016. PMID: 27042289 Free PMC article.
-
Regulatory interactions between androgens, Hoxb5, and TGF β signaling in murine lung development.Biomed Res Int. 2013;2013:320249. doi: 10.1155/2013/320249. Epub 2013 Sep 3. Biomed Res Int. 2013. PMID: 24078914 Free PMC article.
-
Glucocorticoid metabolism in the developing lung: adrenal-like synthesis pathway.J Steroid Biochem Mol Biol. 2013 Nov;138:72-80. doi: 10.1016/j.jsbmb.2013.03.004. Epub 2013 Mar 26. J Steroid Biochem Mol Biol. 2013. PMID: 23537622 Review.
-
Hormonal and developmental regulation of pulmonary surfactant synthesis in fetal lung.Baillieres Clin Endocrinol Metab. 1990 Jun;4(2):351-78. doi: 10.1016/s0950-351x(05)80055-2. Baillieres Clin Endocrinol Metab. 1990. PMID: 2248600 Review.
Cited by
-
MicroRNA Signatures Associated with Bronchopulmonary Dysplasia Severity in Tracheal Aspirates of Preterm Infants.Biomedicines. 2021 Mar 5;9(3):257. doi: 10.3390/biomedicines9030257. Biomedicines. 2021. PMID: 33807742 Free PMC article.
-
Risk factors for respiratory distress syndrome in full-term neonates.Yeungnam Univ J Med. 2018 Dec;35(2):187-191. doi: 10.12701/yujm.2018.35.2.187. Epub 2018 Dec 31. Yeungnam Univ J Med. 2018. PMID: 31620592 Free PMC article.
-
High-risk Factors of Respiratory Distress Syndrome in Term Neonates: A Retrospective Case-control Study.Balkan Med J. 2014 Mar;31(1):64-8. doi: 10.5152/balkanmedj.2014.8733. Epub 2014 Mar 1. Balkan Med J. 2014. PMID: 25207170 Free PMC article.
-
Single-cell analysis reveals differential regulation of the alveolar macrophage actin cytoskeleton by surfactant proteins A1 and A2: implications of sex and aging.Biol Sex Differ. 2016 Mar 18;7:18. doi: 10.1186/s13293-016-0071-0. eCollection 2016. Biol Sex Differ. 2016. PMID: 26998217 Free PMC article.
-
Effects of 5-alpha reductase inhibitors on lung function: A reason for discontinuation during COVID-19 pandemic?Dermatol Ther. 2020 Jul;33(4):e13535. doi: 10.1111/dth.13535. Epub 2020 May 20. Dermatol Ther. 2020. PMID: 32383237 Free PMC article. No abstract available.
References
-
- Fleisher B, Kulovich MV, Hallman M, Gluck L. Lung profile: sex differences in normal pregnancy. Obstet Gynecol. 1985;66(3):327–330. - PubMed
-
- Perelman RH, Palta M, Kirby R, Farrell PM. Discordance between male and female deaths due to the respiratory distress syndrome. Pediatrics. 1986;78(2):238–244. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

